Click on the DOIs or images to read the publications. They are all “open access” unless otherwise noted.
2026
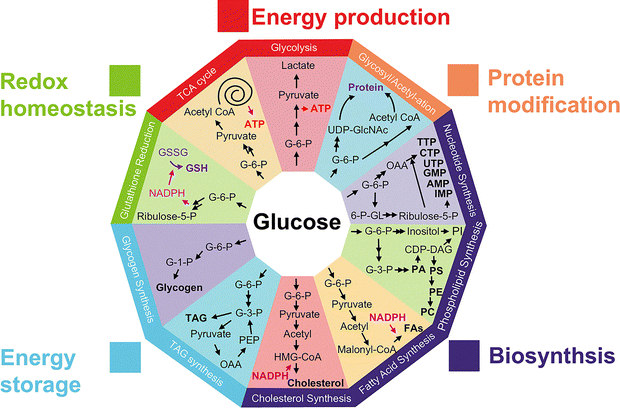
79.) “Design and development of glucose probes for sensing and imaging within biological systems”, Wang, H.; Li, C.; Zhang, F.; Kersch-Hunt, B.; Gardiner, J.E.; Ren, Li, Y.; Lewis, S. E.; Mu, Y.; Yao, Q.; Wu, L.; Groleau, R. R.*; Zhai, W.; Liu, Z.*; James, T. D.*; Anslyn, E. V.*; Wang, K.*, Chem. Soc. Rev., 2026, 55, ASAP, doi:10.1039/D3CS00795B
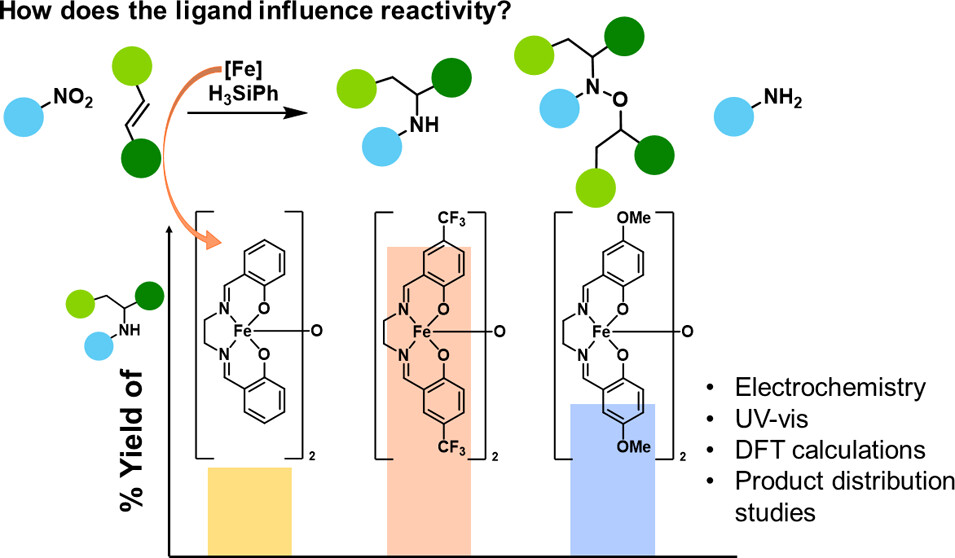
78.) “Effect of the [Fe(salen)]2‑μ-oxo catalyst electronic structure on reductive hydroamination”, Pocock, E.; Buxton, N. J.; Diefenbach, M.; Bond, A. D.; Lewis, S. E.*; Krewald, V.;* Webster, R. L.*, Inorg. Chem., 2026, 65, 999, doi:10.1021/acs.inorgchem.5c05628
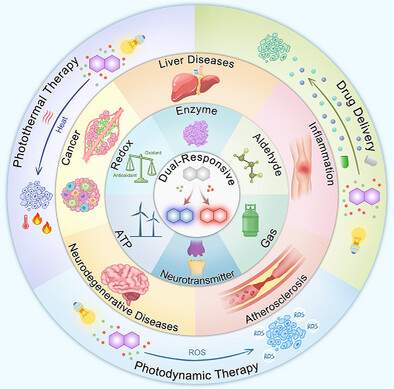
77.) “Dual-responsive fluorescent probes: Advances in biosensing, diagnosis and therapy”, Liu, J.; Wu, L.; Zhu, Z.; Yan, C.; Zhang, Y.; Yang, T.; Xu, S.; Yang, H.; Liu, S.; Tang, W.; Ma, X.; Lewis, S. E.; Wang, Q.*; James, T. D.*; Zhu, W. H.*, Adv. Funct. Mater., 2026, 36, e15602, doi:10.1002/adfm.202515602
2025
76.) “Synthesis and resolution of a 1,1′-biazulene analogue of BINOL”, Gee, A. P.; Gianga, T.-M.; Kociok-Köhn, G.; Pantoş, G. D.; Lewis, S. E.*, RSC Adv., 2025, 15, 14881, doi:10.1039/D5RA02520F

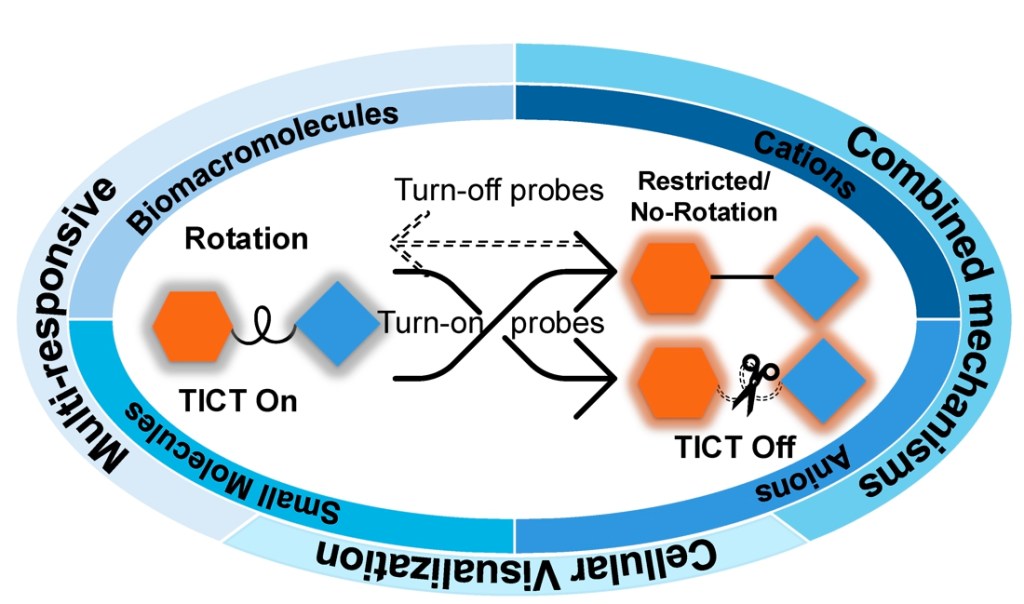
75.) “Twisted intramolecular charge transfer (TICT) based fluorescent probes and imaging agents”, Wu, Y.; Wang, H. M.; Hu, X. L.; Zang, Y.; Li, J.*; Han, H. H.*; He, X. P.*; Lewis, S. E.*; Ismail, H. M.*; James, T. D.*, Chem. Soc. Rev., 2025, 54, 12080, doi:10.1039/D3CS01118F
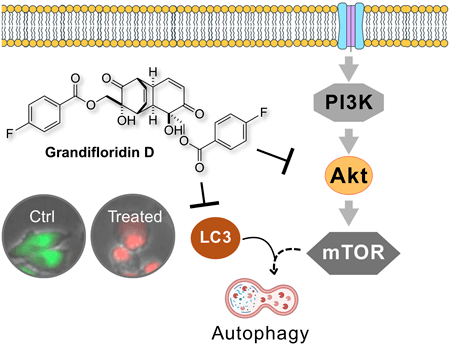
74.) “Grandifloridin D: A Potent antiausterity agent targeting pancreatic cancer cells via Akt/mTOR and autophagy inhibition”, Nguyen, H. H.; Maneenet, J. Fujii, T.; Caggiano, L.; Lewis, S. E.*; Awale, S.*, Biol. Pharm. Bull., 2025, 48, 1784, doi:10.1248/bpb.b25-00545
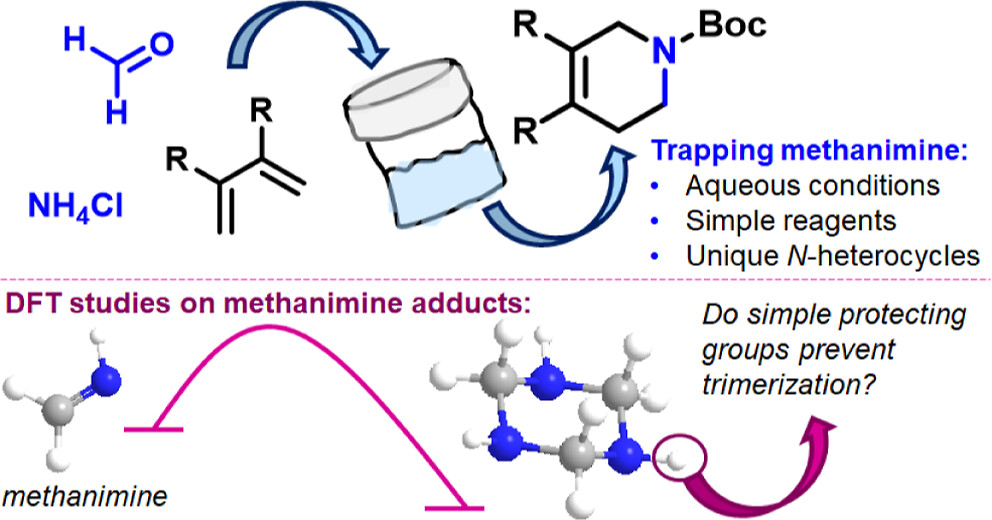
73.) “Intercepting methanimine for the synthesis of piperidine-based N-heterocycles in an aqueous medium”, Pocock, E.; Diefenbach, M.; Hood, T. M.; Nunn, M.; Krewald, V.*; Lewis, S. E.*; Webster, R. L.*, J. Org. Chem., 2025, 90, 11257, doi:10.1021/acs.joc.5c01213
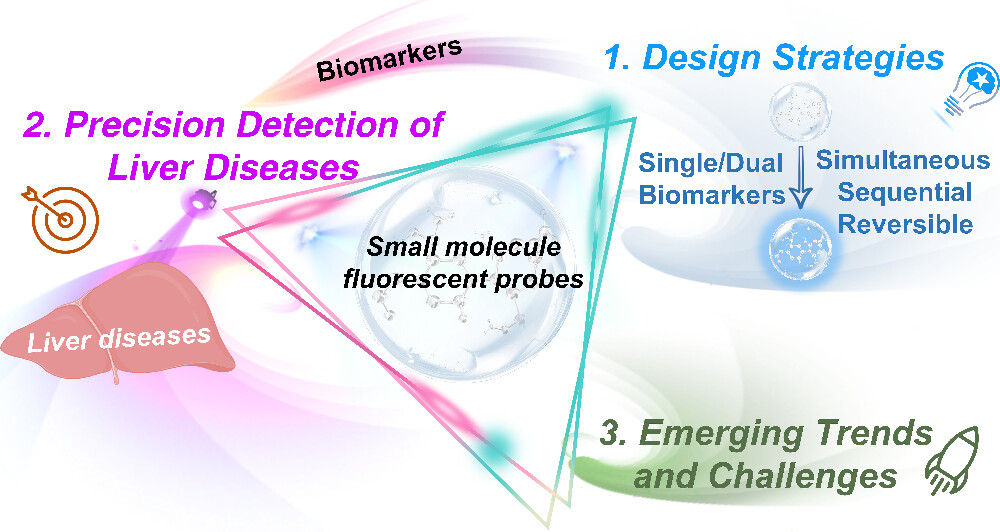
72.) “Advances in organic small molecule-based fluorescent probes for precision detection of liver diseases: A Perspective on emerging trends and challenges”, Wu, L.; Liu, Z.; Wang, K. Groleau, R. R.; Rong, X.; Liu, X.; Liu, C.; Lewis, S. E.; Zhu, B.*; James, T. D.*, J. Am. Chem. Soc., 2025, 147, 9001, doi:10.1021/jacs.4c17092
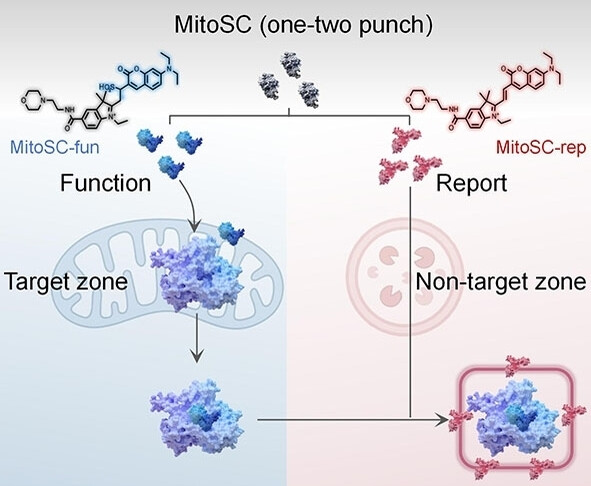
71.) “A Small-molecule drug for the self-checking of mitophagy”, Gao, Y.; Bai, Q.; Ren, Y.; Shao, X.; Zhang, M.; Wu, L.*; Lewis, S. E.; James, T. D.; Chen, X.*; Chen, Q.*, Angew. Chem. Int. Edn., 2025, 64, e202421269, doi:10.1002/anie.202421269
2024
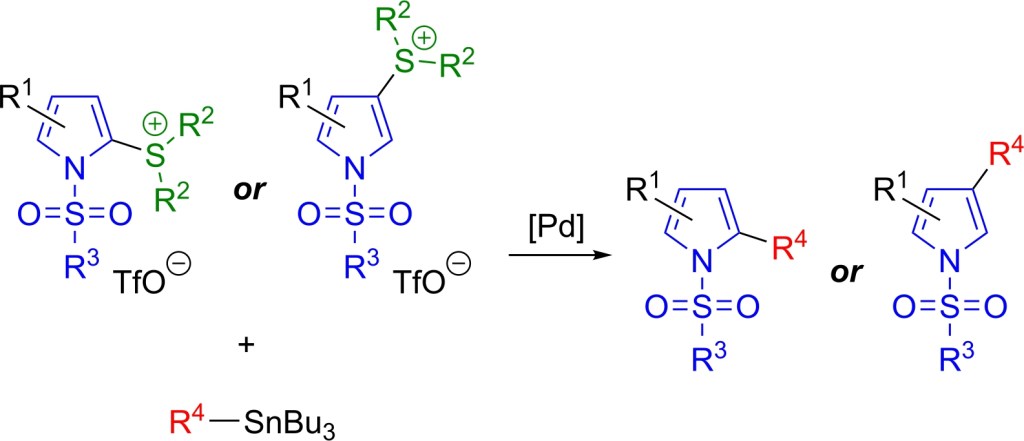
70.) “Pyrrolylsulfonium salts: stable, accessible and versatile pseudohalides for Stille couplings”, Hann, J. L.; Lyall, C. L.; Kociok-Köhn, G.; Lewis, S. E.*, Org. Chem. Front., 2024, 11, 7011, doi:10.1039/D4QO01793E
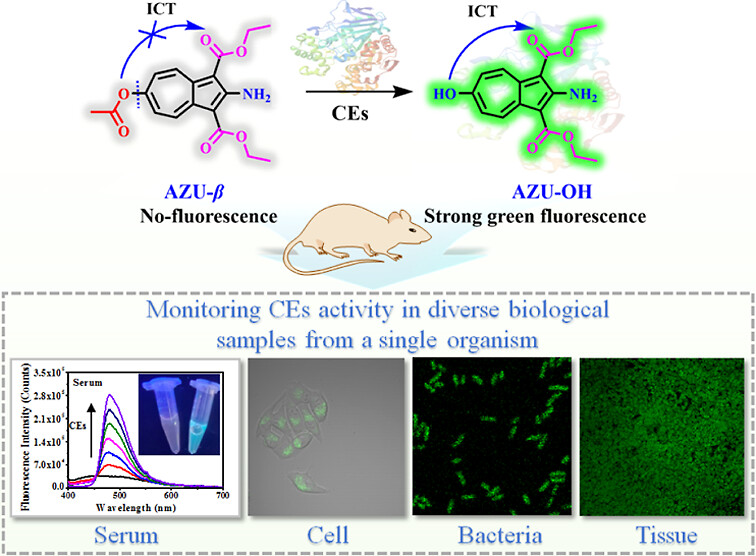
69.) “Amphiphilic azulene-based fluorescent probe for simultaneous monitoring of fluctuations in carboxylesterase activity in diverse biological samples from a single organism”, Cui, Z.; Wang, Y.; Wang, G.; Feng, B.; Lewis, S. E.; Wang, K.; Jiang, K.; James, T. D.*; Zhang, H.*, Anal. Chem., 2024, 96, 19732, doi:10.1021/acs.analchem.4c04926
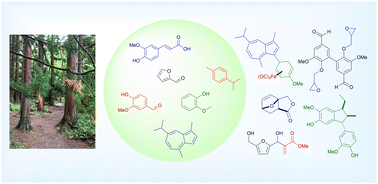
68.) “Carbon–carbon bond formation using aromatics from biomass”, Dunås, P.; Patterson, A. J.; Lewis, S. E.*; Kann, N.*, Chem. Commun., 2024, 60, 14885, doi:10.1039/D4CC05664G
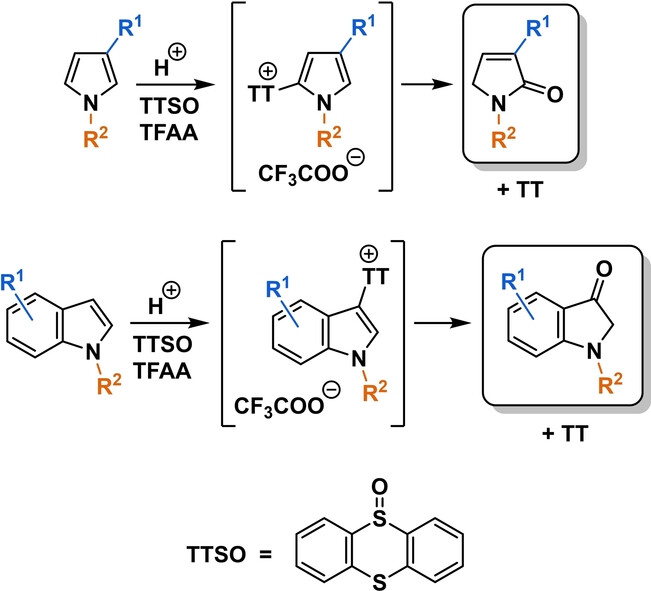
67.) “Unusual regio- and chemoselectivity in oxidation of pyrroles and indoles enabled by a thianthrenium salt intermediate”, Hann, J. L.; Lyall, C. L.; Kociok-Köhn, G.; Faverio, C.; Pantoş, G. D.*; Lewis, S. E.*, Angew. Chem. Int. Edn., 2024, 63, e202405057, doi:10.1002/anie.202405057
66.) “Fluorescent probes for disease diagnosis”, Wang, X., Ding, Q., Groleau, R. R., Wu, L., Mao, Y., Che, F., Kotova, O., Scanlan, E. M., Lewis, S. E.*, Li, P.*, Tang, B.*, James, T. D.*; Gunnlaugsson, T.*, Chem. Rev., 2024, 124, 7106, doi:10.1021/acs.chemrev.3c00776
65.) “Fluorescent small molecule donors”, Chen, G.*; Yu, J.; Wu, L.; Ji, X.; Xu, J.; Wang, C.; Ma, S.; Miao, Q.; Wang, L.; Wang, C.; Lewis, S. E.; Yue, Y.*; Sun, Z.*; Liu, Y.*; Tang, B.*; James, T. D.*, Chem. Soc. Rev., 2024, 53, 6345, doi:10.1039/D3CS00124E
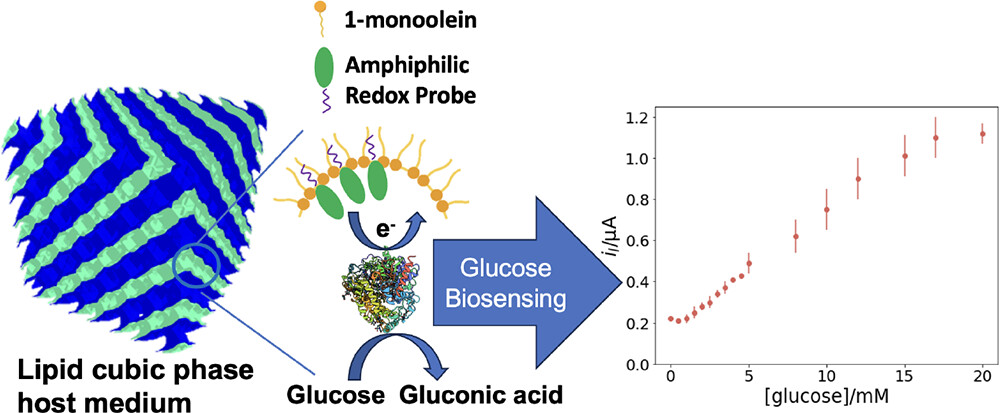
64.) “Development of redox-active lyotropic lipid cubic phases for biosensing platforms”, Liu, W.; Lewis, S. E.; Di Lorenzo, M.; Squires, A. M.*; Langmuir, 2024, 40, 170, doi:10.1021/acs.langmuir.3c02307
2023
63.) “Synthesis of N-alkoxycarbonyl pyrroles from O-substituted carbamates: A synthetically enabling pyrrole protection strategy”, Hann, J. L.; Lyall, C. L. , Kociok-Köhn, G.; Lewis, S. E.*, J. Org. Chem., 2023, 88, 13584, doi:10.1021/acs.joc.3c01257
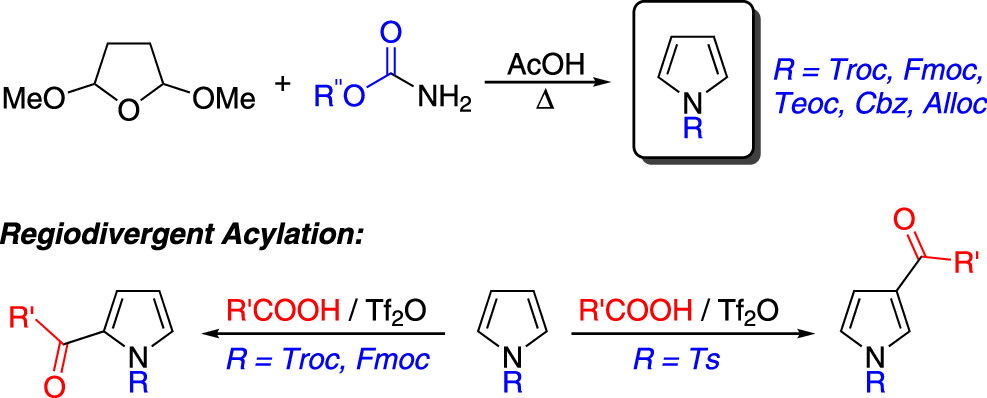
62.) “Photochemical fingerprinting is a sensitive probe for the detection of synthetic cannabinoid receptor agonists; Toward robust point-of-care detection”, Andrews, R.; May, B.; Hernández, F. J.; Townsend, P.; Cozier, G.; Sutcliffe, O. B.; Haines, T. S. F.; Freeman, T.; Scott, J.; Husbands, S.; Blagbrough, I.; Bowman, R.; Lewis, S. E.; Grayson, M.; Crespo-Otero, R.*; Carbery, D.*; Pudney, C.*, Anal. Chem., 2023, 95, 703, doi:10.1021/acs.analchem.2c02529
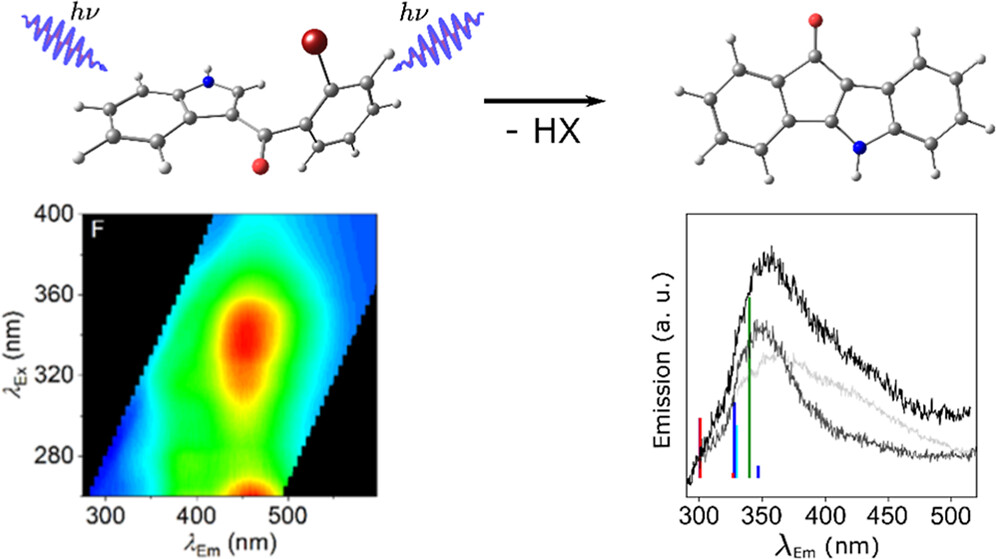
61.) “Persistent azulene α-carbocations: synthesis from aldehydes, spectroscopic and crystallographic properties”, Harabajiu, C.; Hann, J. L; Murfin, L. C., Kociok-Köhn, G.; Lewis, S. E.*, Org. Biomol. Chem., 2023, 21, 858 doi:10.1039/D2OB01695H

2022
60.) “Structural investigation of sulfobetaines and phospholipid monolayers at the air–water interface”, Elstone, N.*; Arnold, T.; Skoda, M. W. A.; Lewis, S. E.; Li, P.; Hazell, G.; Edler, K. J.*, Phys. Chem. Chem. Phys., 2022, 24, 22679, doi:10.1039/d2cp02695c
2021
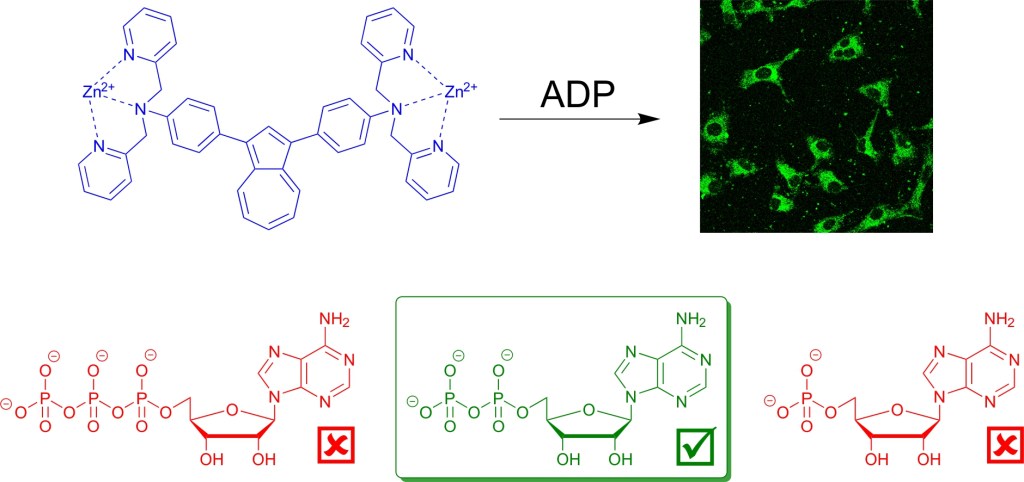
59.) “Azulene-based fluorescent chemosensor for adenosine diphosphate”, López-Alled, C. M.; Park, S. J.; Lee, D. J.; Murfin, L. C.; Kociok-Köhn, G.; Hann, J. L.; Wenk, J.*; James, T. D.*; Kim, H. M.*; Lewis, S. E.*, Chem. Commun., 2021, 57, 10608, doi:10.1039/d1cc04122c
58.) “Palladium-catalyzed stereoselective domino arylation–acylation: an entry to chiral tetrahydrofluorenone scaffolds”, Dunås, P.; Paterson, A. J.; Kociok-Köhn, G.; Rahm, M.; Lewis, S. E.*; Kann, N.*, Chem. Commun., 2021, 57, 6518, doi:10.1039/d1cc02160e

57.) “Azulene – A bright core for sensing and imaging”, Murfin, L. C.*; Lewis, S. E., Molecules, 2021, 26, 353, doi:10.3390/molecules26020353
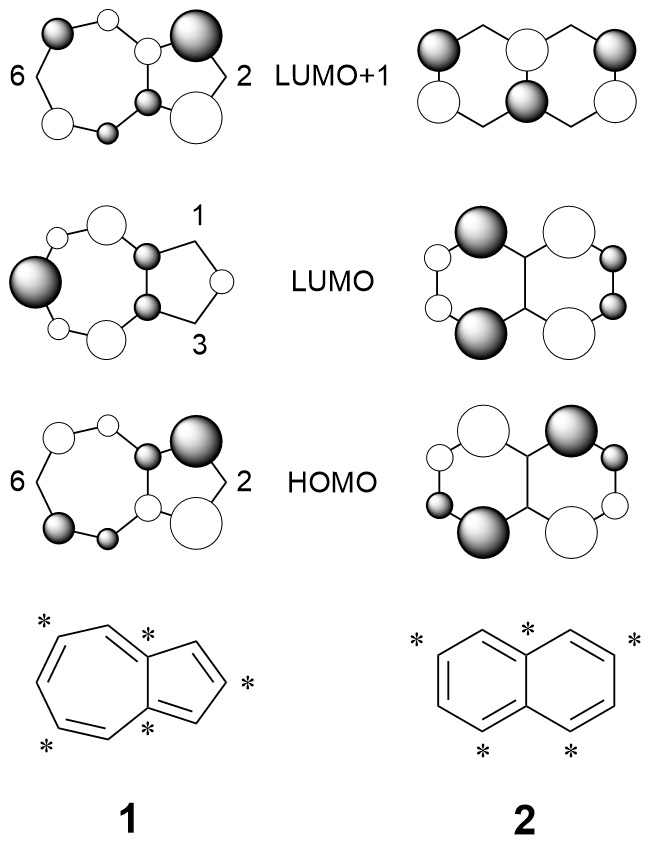
56.) “C4-aldehyde of guaiazulene: synthesis and derivatisation”, Williams, G. E.; Kociok-Köhn, G.; James, T. D.; Lewis, S. E.*, Org. Biomol. Chem., 2021, 19, 2502, doi:10.1039/d0ob02567d
55.) “Fluorescent small organic probes for biosensing”, Tian, X.; Murfin, L. C.; Wu, L.*; Lewis, S. E.*; James, T. D.*, Chem. Sci., 2021, 12, 3406, doi:10.1039/D0SC06928K
2020
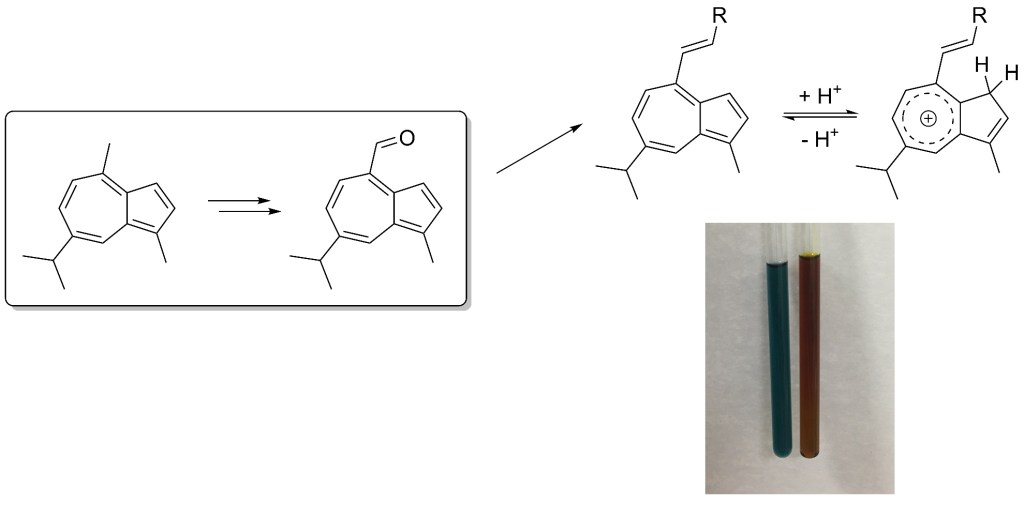
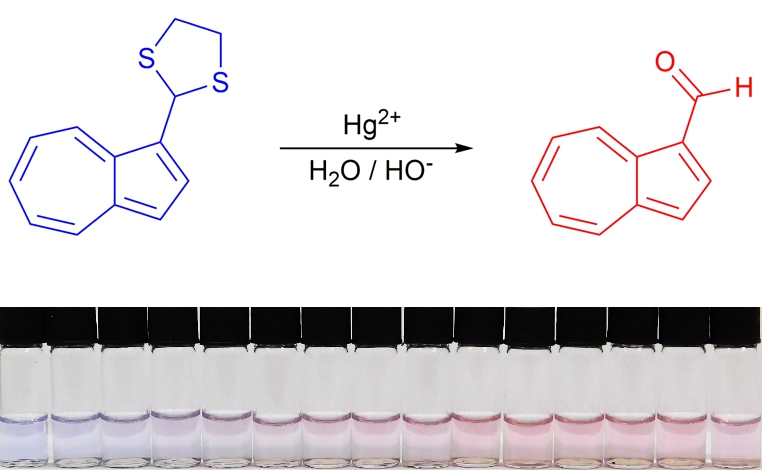
54.) “Colorimetric detection of Hg2+ with an azulene-containing chemodosimeter via dithioacetal hydrolysis”, López-Alled, C. M.; Murfin, L. C.; Kociok-Köhn, G.; James, T. D.*; Wenk, J.*; Lewis, S. E.*, Analyst, 2020, 145, 6262, doi:10.1039/d0an01404d
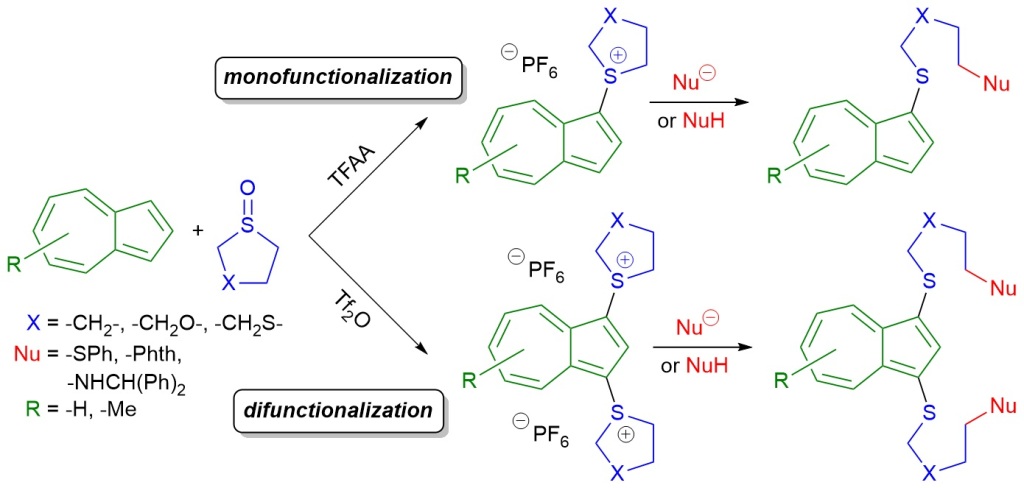
53.) “Azulenesulfonium and azulenebis(sulfonium) salts: Formation by interrupted Pummerer reaction and subsequent derivatisation by nucleophiles”, López-Alled, C. M.; Martin, F. J. O.; Chen, K.-Y.; Kociok-Köhn, G.; James, T. D.; Wenk, J.; Lewis, S. E.*, Tetrahedron, 2020, 76, 131700, doi:10.1016/j.tet.2020.131700
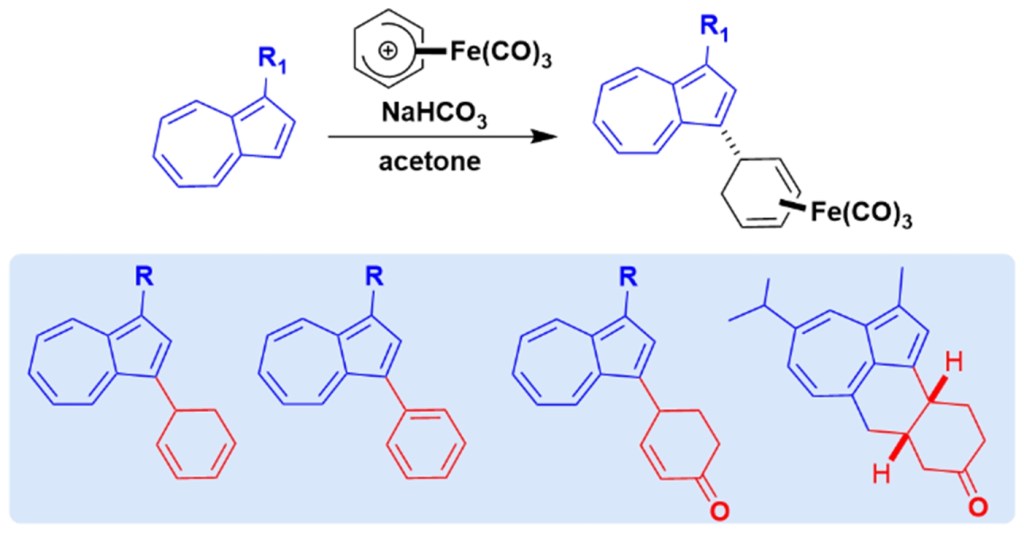
52.) “Azulene functionalization by iron-mediated addition to a cyclohexadiene scaffold”, Dunås, P.; Murfin, L. C.; Nilsson, O. J.; Jame, N.; Lewis, S. E.*; Kann, N.*, J. Org. Chem., 2020, 85, 13453, doi:10.1021/acs.joc.0c01412
51.) “Palladium catalyzed stereoselective arylation of biocatalytically derived cyclic 1,3-dienes: Chirality transfer via a Heck-type mechanism”, Paterson, A. J.; Dunås, P.; Rahm, M.; Norrby, P.-O.; Kociok-Köhn, G.; Lewis, S. E.*; Kann, N.*, Org. Lett., 2020, 22, 2464, doi:10.1021/acs.orglett.0c00708
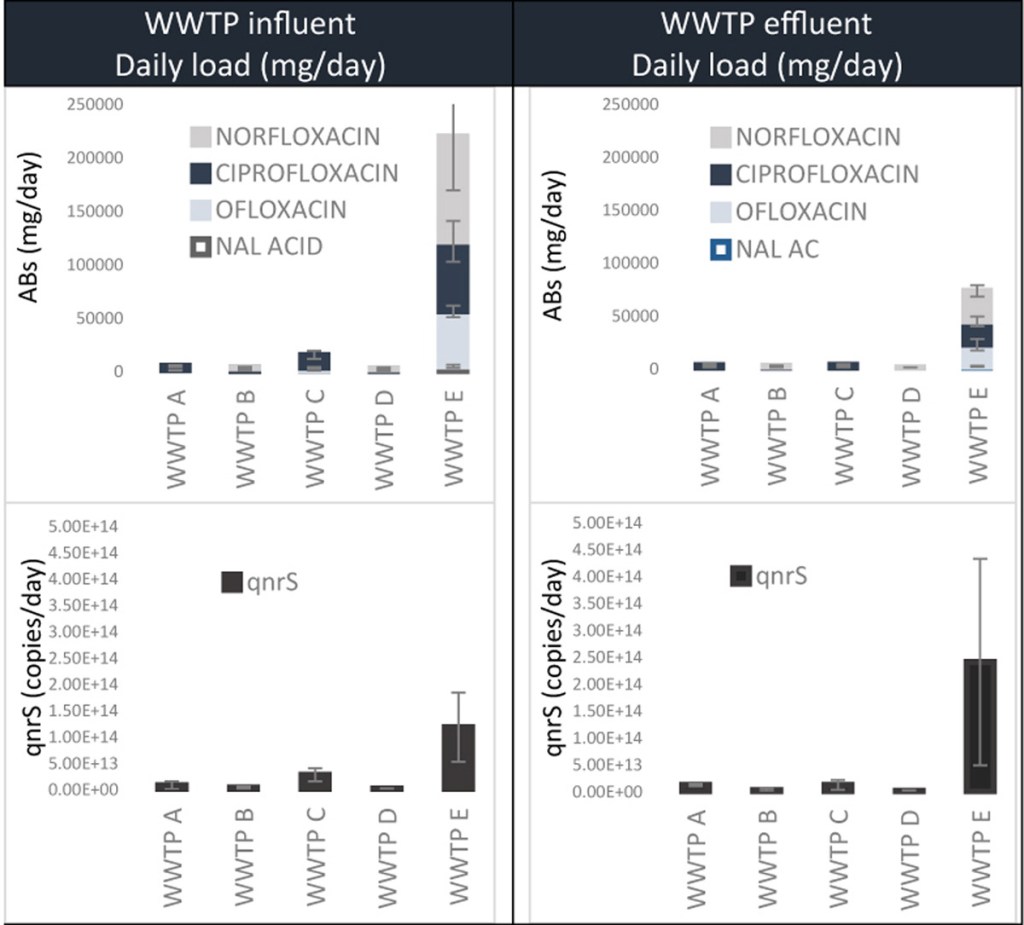
50.) “(Fluoro)quinolones and quinolone resistance genes in the aquatic environment: A river catchment perspective”, Castrignanò, E.; Kannan, A. M.; Proctor, K.; Petrie, B.; Hodgen, S.; Feil, E. J.; Lewis, S. E.; Lopardo, L.; Camacho-Muñoz, D.; Rice, J.; Cartwright, N.; Barden, R.; Kasprzyk-Hordern, B.*, Water Res., 2020, 182, 116015, doi:10.1016/j.watres.2020.116015
49.) “A Colorimetric chemosensor based on a Nozoe azulene that detects fluoride in aqueous/alcoholic media”, Murfin, L. C.; Chiang, K. X. C.; Lyall, C. L.; Williams, G. T.; Wenk, H.*; Jenkins, T.*; James, T. D.*; Lewis, S. E.*, Front. Chem., 2020, doi: 10.3389/fchem.2020.00010

48.) “A simple, azulene-based colorimetric probe for the detection of nitrite in water”, Murfin, L. C.; López-Alled, C. M.; Sedgwick, A. C.; Wenk, J.*; James, T. D.*; Lewis, S. E.*, Front. Chem. Sci. Eng., 2020, doi:10.1007/s11705-019-1790-7

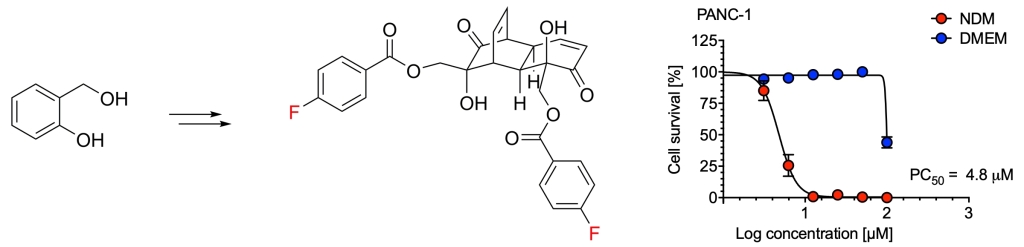
47.) “Sidechain Diversification of Grandifloracin Allows Identification of Analogues with Enhanced Anti‐Austerity Activity against Human PANC‐1 Pancreatic Cancer Cells”, Alexander, B. E.; Sun, S.; Palframan, M. J.; Kociok‐Köhn, G.; Dibwe, D. F.; Watanabe, S.; Caggiano, L.*; Awale, S.*; Lewis, S. E.*, ChemMedChem, 2020, 15, 125, doi:10.1002/cmdc.201900549
2019
46.) “Azulene-Derived Fluorescent Probe for Bioimaging: Detection of Reactive Oxygen and Nitrogen Species by Two-Photon Microscopy”, Murfin, L. C.; Weber, M.; Park, S. J.; Kim, W. T.; Lopez-Alled, C. M.; McMullin, C. L.; Pradaux-Caggiano, F.; Lyall, C. L.; Kociok-Köhn, G.; Wenk, J. H.; Bull, S. D.; Yoon, J.; Kim, H. M.*; James, T. D.*; Lewis, S. E.*, J. Am. Chem. Soc. 2019, 141, 19389, doi: 10.1021/jacs.9b09813

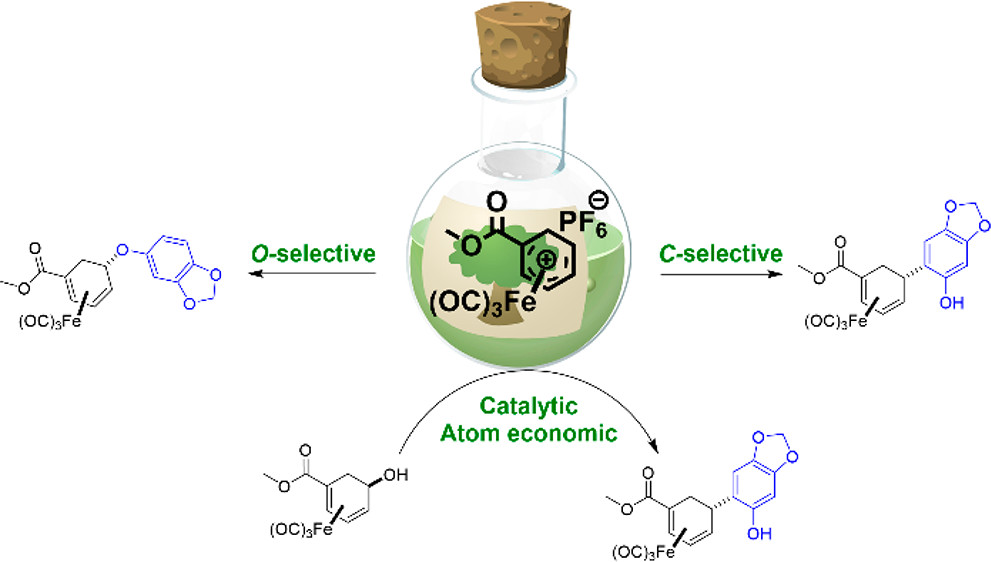
45.) “Selective Iron-Mediated C- and O-Addition of Phenolic Nucleophiles to a Cyclohexadiene Scaffold Using Renewable Precursors”, Dunås, P.; Paterson, A. J.; Kociok-Köhn, G.; Lewis, S. E.*; Kann, N.*, ACS Sustainable Chem. Eng., 2019, 7, 7155, doi:10.1021/acssuschemeng.9b00127
44.) “Partial Cation Substitution Reduces Iodide Ion Transport in Lead Iodide Perovskite Solar Cells”, Ferdani, D.; Pering, S.; Ghosh, D.; Kubiak, P.; Walker, A.; Lewis, S. E.; Johnson, A. L.; Baker, P. J.; Islam, M. S.; Cameron, P. J., Energy Environ. Sci., 2019, 12, 2264, doi: 10.1039/c9ee00476a

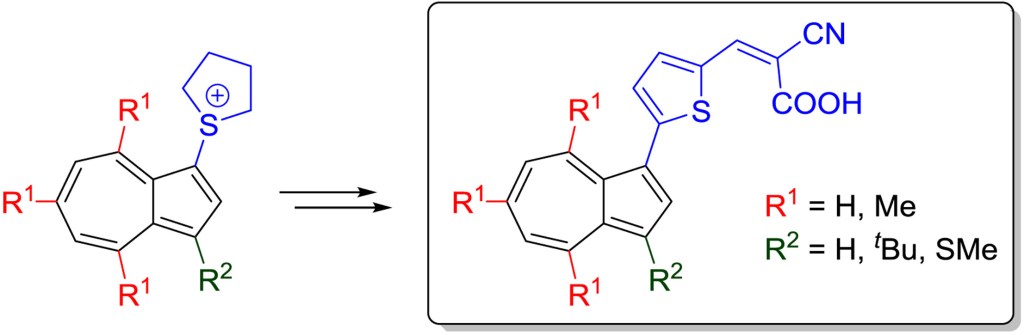
43.) “Azulenes with aryl substituents bearing pentafluorosulfanyl groups: synthesis, spectroscopic and halochromic properties”, Webster, S. J.; López-Alled, C. M.; Liang, X.; McMullin, C. L.; Kociok-Köhn, G.; Lyall, C. L.; James, T. D.; Wenk, J.; Cameron, P. J.; Lewis, S. E.*, New J. Chem., 2019, 43, 992, doi:10.1039/C8NJ05520C

2018
42.) “Azulene–Thiophene–Cyanoacrylic acid dyes with donor-π-acceptor structures. Synthesis, characterisation and evaluation in dye-sensitized solar cells”, Cowper, P*; Pockett, A.; Kociok-Köhn, G.; Cameron, P. J.; Lewis, S. E*, Tetrahedron, 2018, 74, 2775, doi:10.1016/j.tet.2018.04.043
41.) “Self-assembly and surface behaviour of pure and mixed zwitterionic amphiphiles in a deep eutectic solvent”, Sanchez-Fernandez, A.; Moody, G. L.; Murfin, L. C.; Arnold,T.; Jackson, A. J.; King, S. M.; Lewis, S. E.; Edler, K. J., Soft Matter, 2018, 14, 5525-5536. doi:10.1039/c8sm00755a
2017
40.) “Enantioselective transformation of fluoxetine in water and its ecotoxicological relevance”, Andrés-Costa, M. J.; Proctor, K.; Sabatini, M. T.; Gee, A. P.; Lewis, S. E.; Pico, Y.; Kasprzyk-Hordern, B., Sci. Rep., 2017, 7, 15777. doi:10.1038/s41598-017-15585-1

39.) “Azetidinium lead iodide for perovskite solar cells”, Pering, S. R.; Deng, W.; Troughton, J. R.; Kubiak, P. S.; Ghosh, D.; Niemann, R. G.; Brivio, F.; Jeffrey, F. E.; Walker, A. B.; Islam, M. S.; Watson, T. M.; Raithby, P. R.; Johnson, A. L.; Lewis, S. E.; Cameron; P. J.*, J. Mater. Chem. A, 2017,5, 20658-20665. doi:10.1039/C7TA07545F
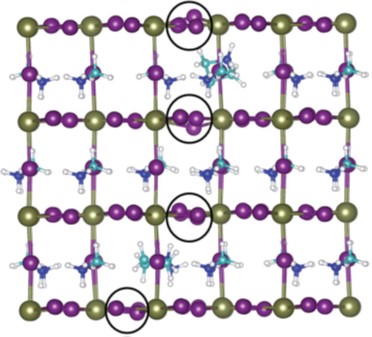
38.) “Biocatalytic dearomatisation of para-fluorobenzoic acid – Access to versatile homochiral building blocks with quaternary centres”, Nash, T. J.; Wharry, S.; Moody, T. S.*; Lewis, S. E.*, Chimica Oggi, 2017, 35, 90, http://www.teknoscienze.com/tks_article/biocatalytic-dearomatisation-of-para-fluorobenzoic-acid-access-to-versatile-homochiral-building-blocks-with-quaternary-centres/
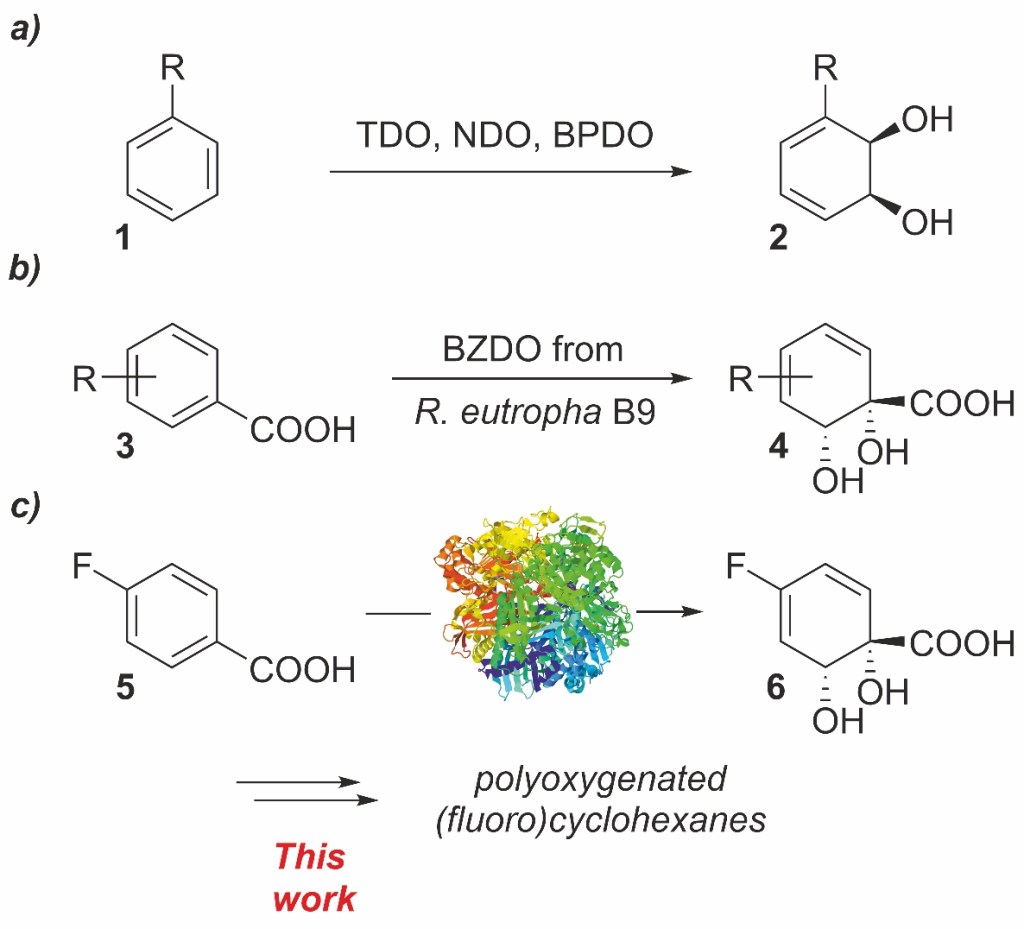
37.) “Azulene–boronate esters: colorimetric indicators for fluoride in drinking water”, López-Alled, C. M.; Sanchez-Fernandez, A.; Edler, K. J.; Sedgwick, A. C.; Bull, S. D.; McMullin, C. L.; Kociok-Köhn, G.; James, T. D.*; Wenk, J.*; Lewis, S. E.*, Chem. Commun., 2017, 53, 12580, doi:10.1039/c7cc07416f
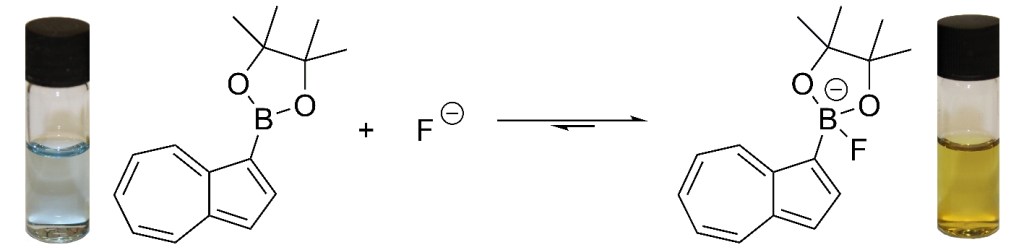
36.) “Phosphorus-substituted azulenes accessed via direct Hafner reaction of a phosphino cyclopentadienide”, Gee, A. P.; Cosham, S. D.; Johnson, A. L.; Lewis, S. E.* Synlett, 2017, 973, doi:10.1055/s-0036-1589936
2016
35.) “What difference does a thiophene make? Evaluation of a 4,4’-bis(thiophene) functionalised 2,2’-bipyridyl copper(I) complex in a dye-sensitized solar cell”, Wills, K. A.; Mandujano-Ramírez, H. A.; Merino, G.; Oskam, G.; Cowper, P.; Jones, M. D.*; Cameron, P. J.*; Lewis, S. E.* Dyes and Pigments, 2016, 134, 419, doi:10.1016/j.dyepig.2016.07.023
34.) “Asymmetric Dearomatization Under Enzymatic Conditions”, Lewis, S. E. in Asymmetric Dearomatization Reactions (You, S.-L. ed.) Wiley-VCH, 2016, pp 279–346. doi:10.1002/9783527698479.ch12
33.) “Langmuir monolayers composed of single and double tail sulfobetaine lipids”, Hazell, G.; Gee, A. P.; Arnold, T.; Edler, K. J.*; Lewis, S. E. J. Colloid Interface Sci., 2016, 474, 190, doi:10.1016/j.jcis.2016.04.020
32.) “Azulenesulfonium Salts: Accessible, Stable and Versatile Reagents for Cross-Coupling”, Cowper, P.*; Jin, Y.; Turton, M. D.; Kociok-Köhn, G.; Lewis, S. E.* Angew. Chem. Int. Edn., 2016, 55, 2564, doi:10.1002/anie.201510666
2015
31.) “Biotransformations of Arenes: An Overview”, Lewis, S. E. in Arene Chemistry: Reaction Mechanisms and Methods for Aromatic Compounds (Mortier, J. ed.) Wiley-VCH, 2015, pp 915–937. doi:10.1002/9781118754887.ch32
30.) “Tricarbonyliron(0) complexes of bio-derived η4 cyclohexadiene ligands: An approach to analogues of oseltamivir”, ten Broeke, M.; Ali Khan, M.; Kociok-Köhn, G.; Kann, N.; Lewis, S. E.* J. Organomet. Chem., 2015, 799-800, 19, doi:10.1016/j.jorganchem.2015.09.005
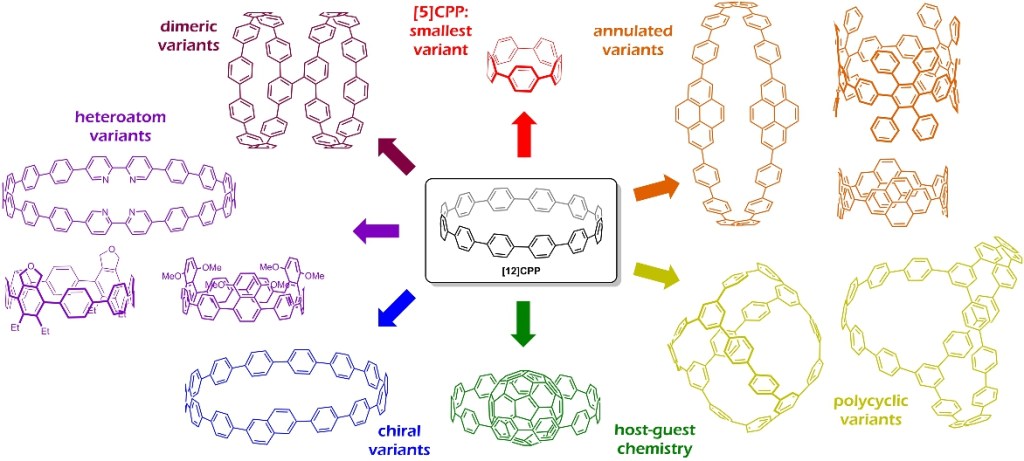
29.) “Cycloparaphenylenes and related nanohoops”, Lewis, S. E.* Chem. Soc. Rev., 2015, 44, 2221–2304. doi:10.1039/c4cs00366g
2014
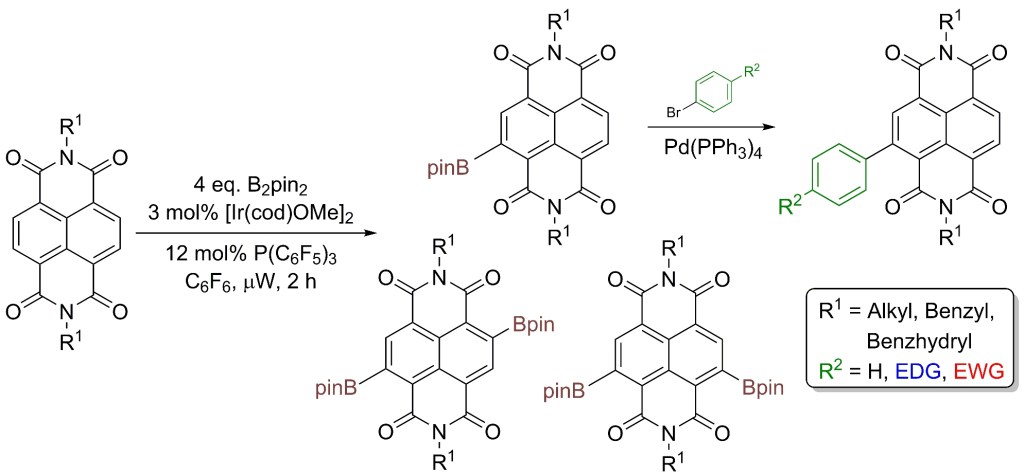
28.) “Direct core functionalisation of naphthalenediimides by iridium catalysed C–H borylation”, Lyall, C. L.; Shotton, C. C.; Pérez-Salvia, M.; Pantoş, G. D.*; Lewis, S. E.* Chem. Commun., 2014, 50, 13837, doi:10.1039/c4cc06522k
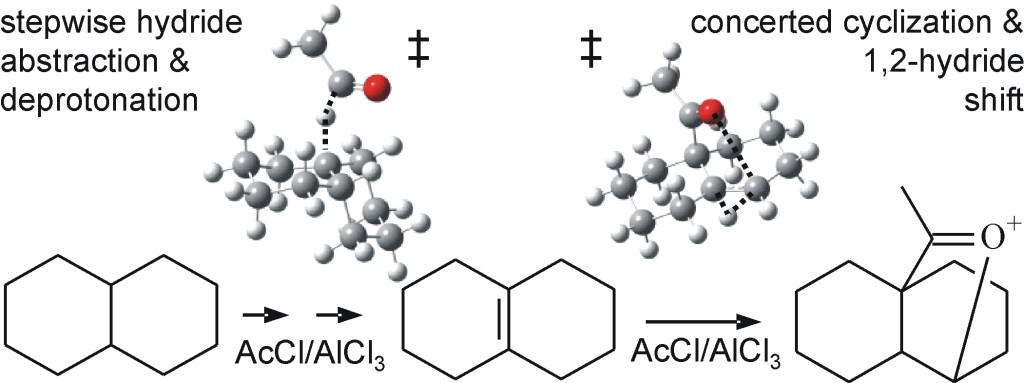
27.) “C–H functionalization of sp3 centers with aluminum: a computational and mechanistic study of the Baddeley reaction of decalin”, Lyall,C. L.; Sato, M.; Uosis-Martin, M.; Asghar, S. F.; Jones, M. D.; Williams, I. H.*; Lewis, S. E.* J. Am. Chem. Soc., 2014, 136, 13745, doi:10.1021/ja5062246
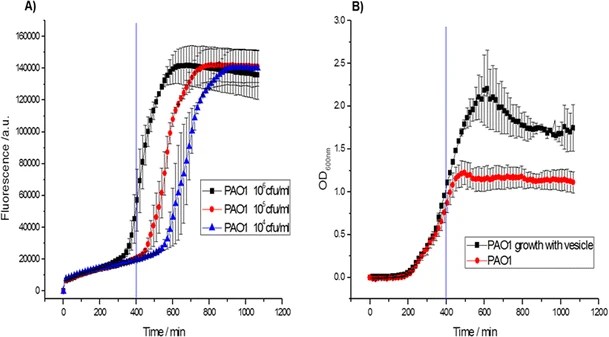
26.) “A new assay for rhamnolipid detection—important virulence factors of Pseudomonas aeruginosa”, Laabei, M.; Jamieson, W. D.; Lewis, S. E.; Diggle, S. P.; Jenkins, A. T. A.* Appl. Microbiol. Biotech., 2014, 98, 7199, doi:10.1007/s00253-014-5904-3
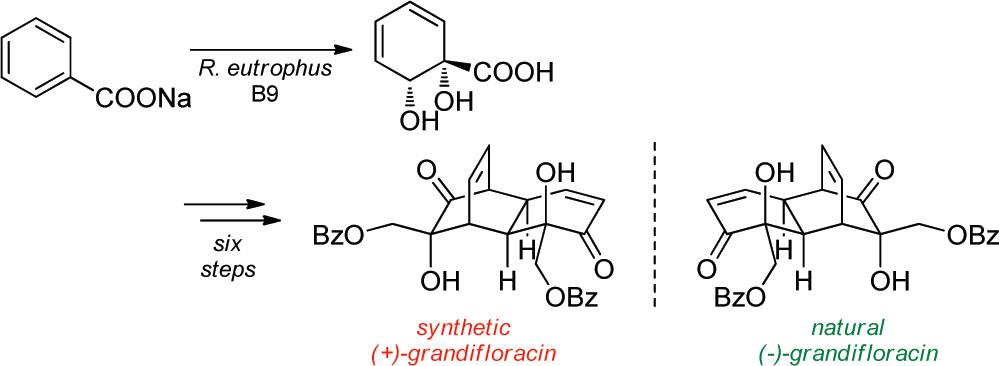
25.) “The Enone Motif of (+)-Grandifloracin is Not Essential for “Anti-Austerity” Antiproliferative Activity”, Ali Khan, M.; Wood, Pauline J.; Lamb-Guhren, N. M.; Caggiano, L.; Kociok-Köhn, G.; Tosh, D.; Lewis, S. E.* Bioorg. Med. Chem. Lett., 2014, 24, 2815, doi:10.1016/j.bmcl.2014.04.111
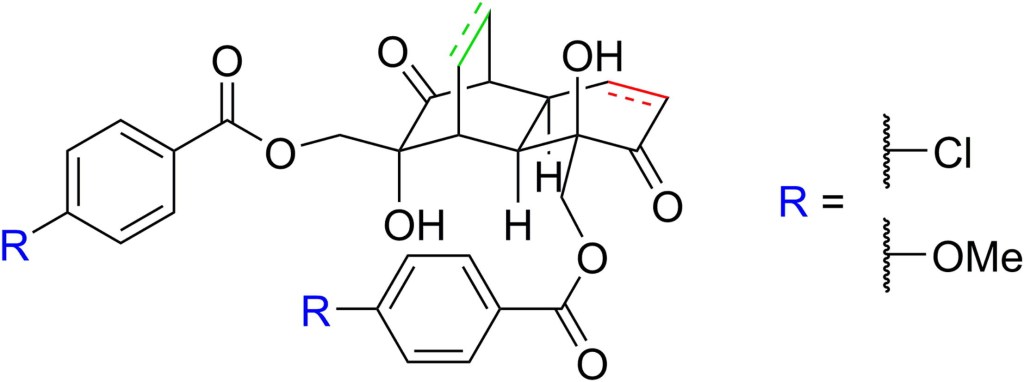
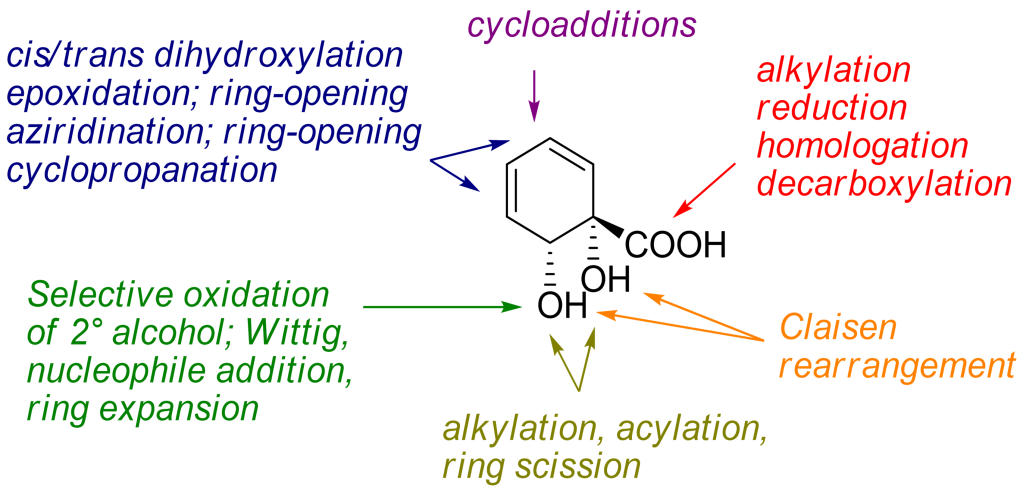
24.) “Applications of biocatalytic arene ipso,orthocis-dihydroxylation in synthesis”, Lewis, S. E.* Chem. Commun., 2014, 50, 2821, doi:10.1039/c3cc49694e
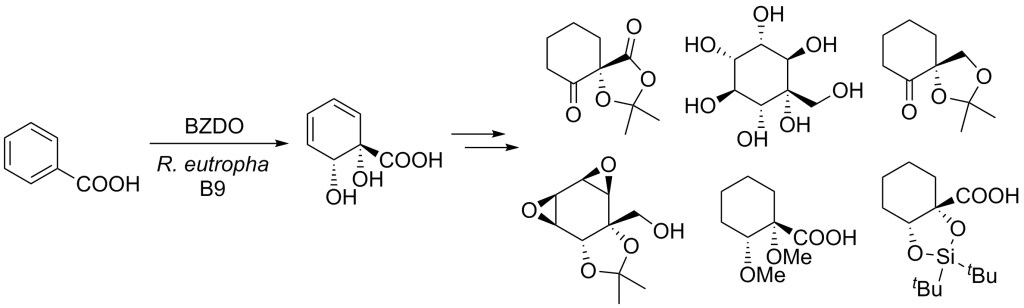
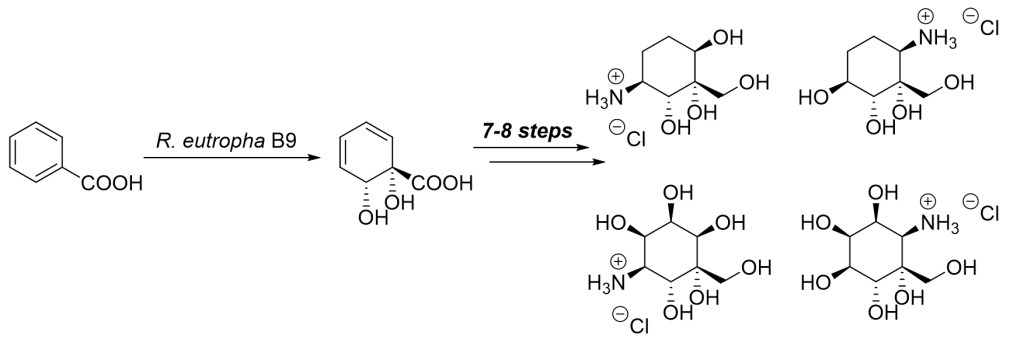
23.) “Benzoate dioxygenase from Ralstonia eutropha B9 – unusual regiochemistry of dihydroxylation permits rapid access to novel chirons”, Griffen, J. A.; Kenwright, S. J.; Abou-Shehada, S.; Wharry, S.; Moody, T. S.*; Lewis, S. E.* Org. Chem. Front., 2014, 1, 79, doi:10.1039/c3qo00057e
2013
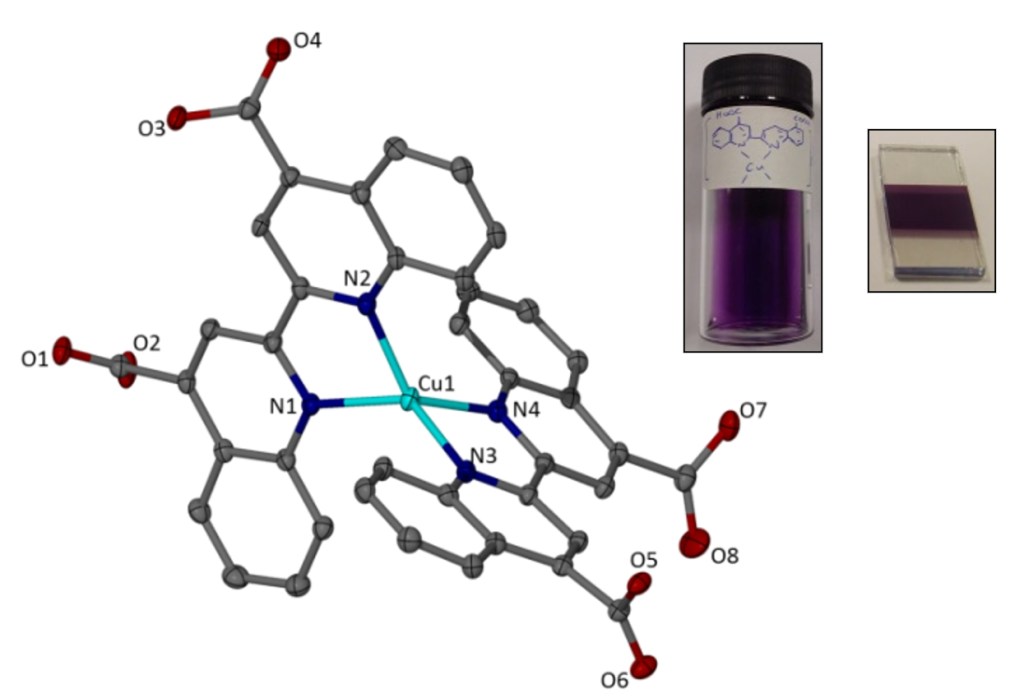
22.) “Investigation of a copper(I) biquinoline complex for application in dye-sensitized solar cells”, Wills, K. A.; Mandujano-Ramírez, H. J.; Merino, G.; Mattia, D.; Hewat, T.; Robertson, N.; Oskam, G.; Jones, M. D.; Lewis, S. E.; Cameron, P. J.*, RSC Advances, 2013, 3, 23361, doi:10.1039/c3ra44936j
21.) “A Model System for the Synthesis of Complanadine Alkaloids by ‘Diverted Kondrat’eva’ Oxazole–Olefin Cycloaddition”, Uosis-Martin, M.; Pantoş, G. D.; Mahon, M. F.; Lewis, S. E.* J. Org. Chem., 2013, 78, 6253, doi:10.1021/jo401014n
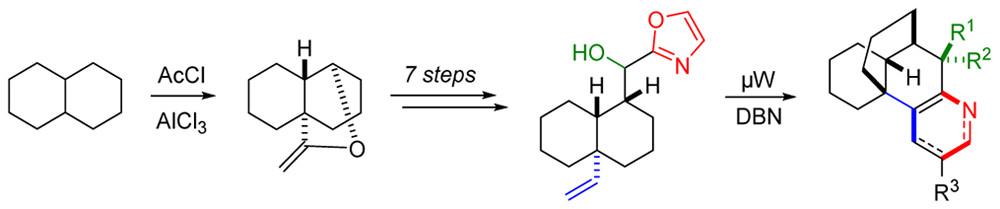
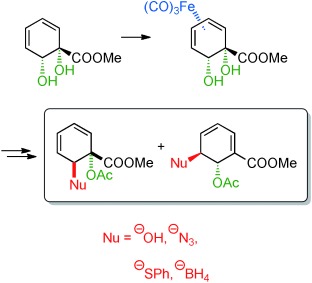
20.) “New Aminocyclitols with Quaternary Stereocentres via Acylnitroso Cycloaddition with an ipso,ortho-Arene Dihydrodiol”, Griffen, J. A.; White, J. C.; Kociok-Köhn, G.; Lloyd, M. D.; Wells, A.; Arnot, T. C.; Lewis, S. E.* Tetrahedron, 2013, 69, 5989, doi:10.1016/j.tet.2013.04.033
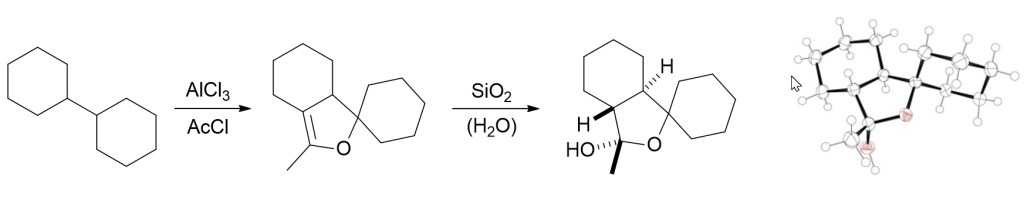
19.) “Aliphatic C–H Activation with Aluminium Trichloride–Acetyl Chloride: Expanding the Scope of the Baddeley Reaction for the Functionalisation of Saturated Hydrocarbons”, Lyall, C. L.; Uosis-Martin, M.; Lowe, J. P.; Mahon, M. F.; Pantoş, G. D.; Lewis, S. E.* Org. Biomol. Chem., 2013, 11, 1468, doi:10.1039/c2ob26765a
2012
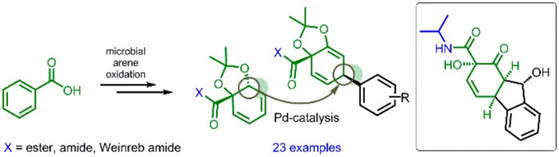
18.) “Valuable New Cyclohexadiene Building Blocks via Cationic η5 Iron Carbonyl Complexes Derived from a Microbial Arene Oxidation Product”, Ali Khan, M.; Mahon, M. F.; Lowe, J. P.; Stewart, A. J. W.; Lewis, S. E.* Chem. Eur. J., 2012, 18, 13480, doi:10.1002/chem.201202411
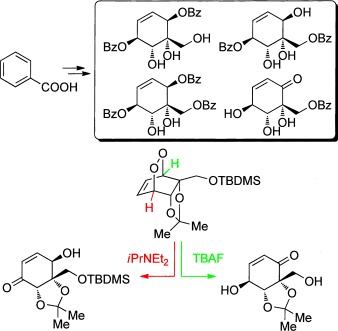
17.) “Photooxygenation of a Microbial Arene Oxidation Product and Regioselective Kornblum–DeLaMare Rearrangement. Total Synthesis of Zeylenols and Zeylenones”, Palframan, M. J.; Kociok-Köhn, G.; Lewis, S. E.* Chem. Eur. J., 2012, 18, 4766, doi:10.1002/chem.201104035
2011
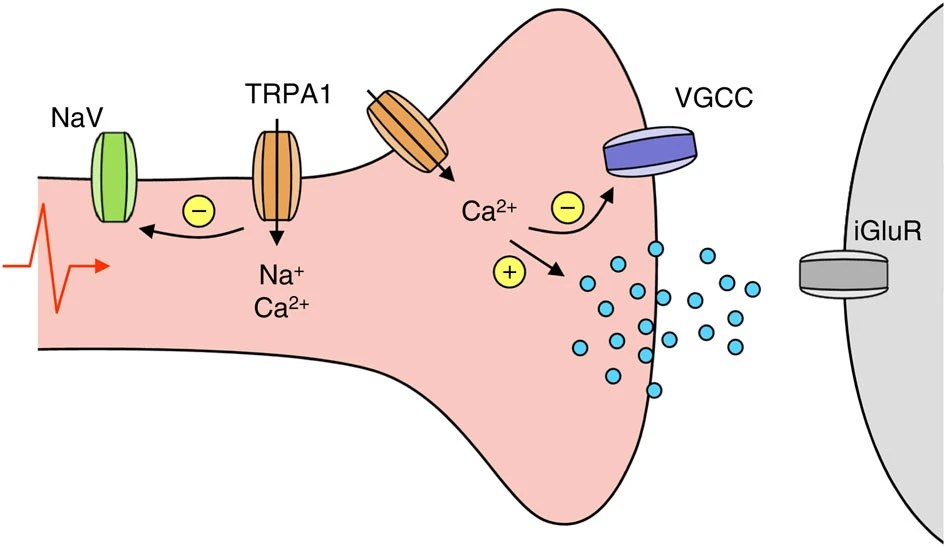
16.) “TRPA1 Mediates Spinal Antinociception Induced by Acetaminophen and the Cannabinoid Δ9-Tetrahydrocannabiorcol”, Andersson, D. A.; Gentry, C.; Alenmyr, L.; Killander, D.; Lewis, S. E.; Andersson, A.; Bucher, B.; Galzi, J.-L.; Sterner, O.; Bevan, S.; Högestätt, E. D.; Zygmunt, P. M. Nature Commun., 2011, 2, 551, doi:10.1038/ncomms1559
15.) “A Cobalt Complex of a Microbial Arene Oxidation Product”, van der Waals, D.; Pugh, T.; Ali Khan, M.; Stewart, A. J. W.; Johnson, A. L.; Lewis, S. E.* Chem. Central J., 2011, 5, 80, doi:10.1186/1752-153X-5-80
14.) “Concise Synthesis of 1,4a-Bifunctionalised Decalin Building Blocks by C–H Activation of Decalin”, Uosis-Martin, M.; Mahon, M. F.; Yevglevskis, M.; Lewis, S. E.* Synlett, 2011, 2211, doi: 10.1055/s-0030-1261184
13.) “Synthetic methods Part (II): oxidation and reduction methods”, Asghar, S. F.; Lewis, S. E.* Annu. Rep. Prog. Chem., Sect. B: Org. Chem., 2011, 107, 34–67, doi:10.1039/c1oc90012a [Not open access]
12.) “Total Synthesis of (+)-Grandifloracin by Iron Complexation of a Microbial Arene Oxidation Product”, Palframan, M. J.; Kociok-Köhn, G.; Lewis, S. E.* Org. Lett., 2011, 13, 3150, doi:10.1021/ol201057r
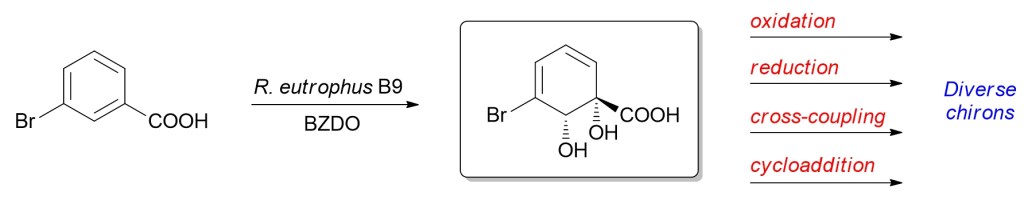
11.) “Expanding the chiral pool: oxidation of meta-bromobenzoic acid by R. eutrophusB9 allows access to new reaction manifolds”, Griffen, J. A.; Le Coz, A. M.; Kociok-Köhn, G.; Ali Khan, M.; Stewart, A. J. W; Lewis, S. E.* Org. Biomol. Chem., 2011, 9, 3920, doi:10.1039/c1ob05131h
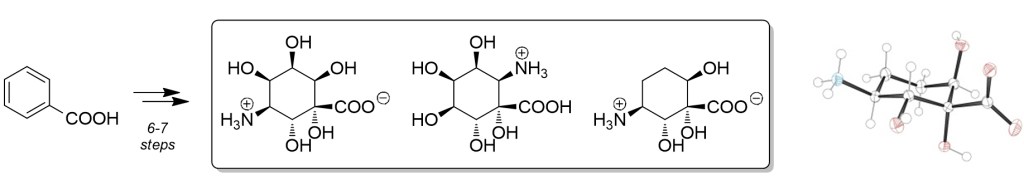
10.) “Inosaminoacids: novel inositol–amino acid hybrid structures accessed by microbial arene oxidation”, Pilgrim, S.; Kociok-Köhn, G.; Lloyd, M. D.; Lewis, S. E.* Chem. Commun., 2011, 47, 4799, doi:10/1039/c1cc10643k
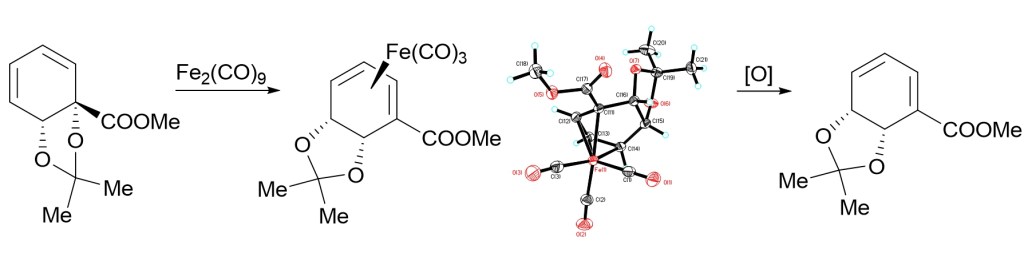
9.) “Accessing the antipodal series in microbial arene oxidation: a novel diene rearrangement induced by tricarbonyliron(0) complexation“, Ali Khan, M.; Lowe, J. P.; Johnson, A. L.; Stewart, A. J. W.; Lewis, S. E.* Chem. Commun., 2011, 47, 215, doi:10.1039/c0cc01169j
2010
8.) “Iron(0)tricarbonyl Complexes of Microbially-Derived Cyclohexadiene Ligands Containing Quaternary Stereocenters”, Ali Khan, M.; Mahon, M. F.; Stewart, A. J. W.; Lewis, S. E.* Organometallics, 2010, 29, 199, doi:10.1021/om9009069 [Not open access]
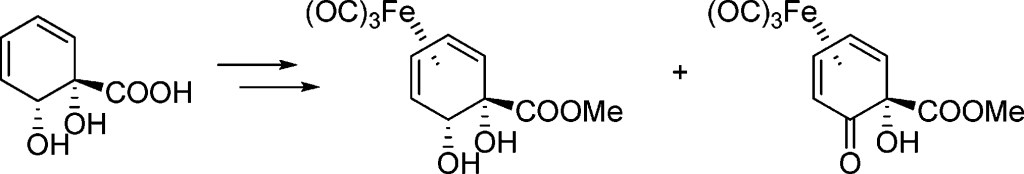
7.) “Transannular decarboxylative Claisen rearrangement reactions for the synthesis of sulfur-substituted vinylcyclopropanes”, Craig, D.*; Gore, S. J.; Lansdell, M. I.; Lewis, S. E.; Mayweg, A. V. M.; White, A. J. P. Chem. Commun., 2010, 46, 4991, doi:10.1039/c0cc00976h [Not open access]
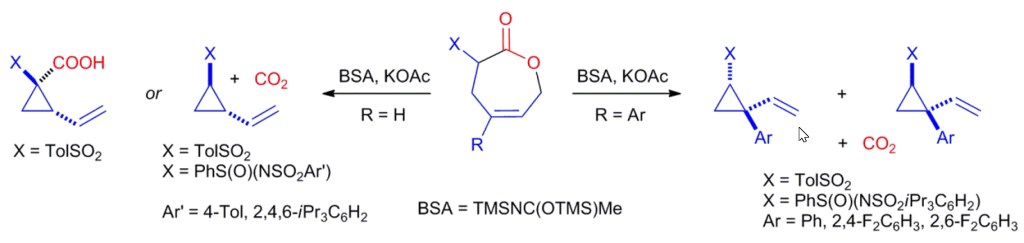
6.) “Synthesis and Characterisation of Novel Alkane-α,ω-diyl bis(silyl triflates)”, Baker, T.; Lewis, S. E.* Synth. Commun., 2010, 40, 2747, doi:10.1080/00397910903318724 [Not open access]
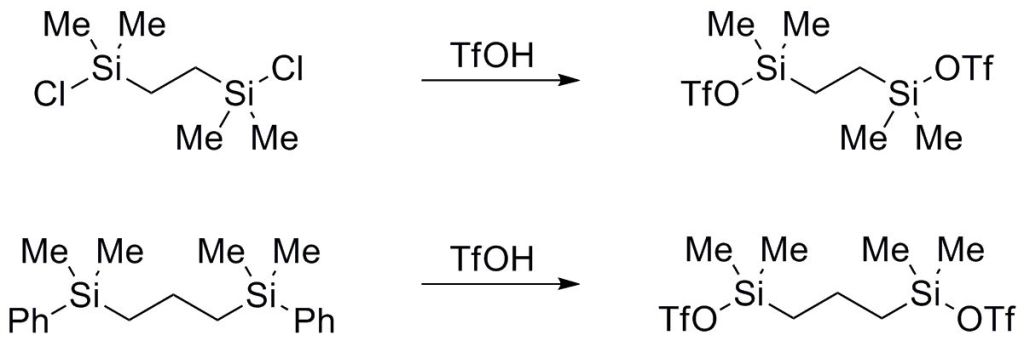
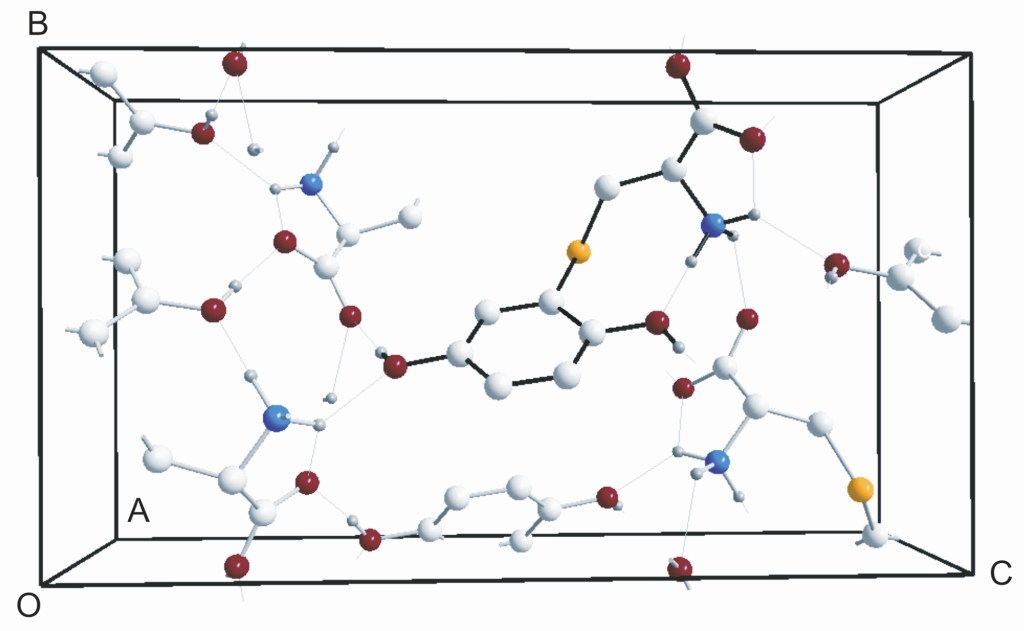
5.) “Crystallographic rationalization of the reactivity and spectroscopic properties of (2R)-S-(2,5-dihydroxyphenyl)cysteine”, Kociok-Köhn, G.; Lewis, S. E.* Acta Cryst., 2010, C66, o187, doi:10.1107/S0108270110005780
4.) “Synthetic methods: Part (ii) Oxidation and reduction methods”, Lewis, S. E.* Annu. Rep. Prog. Chem., Sect. B: Org. Chem., 2010, 106, 34–75. doi:10.1039/b927087f [Not open access]
2009
3.) “Synthetic methods: Part (ii) Oxidation and reduction methods”, Lewis, S. E.* Annu. Rep. Prog. Chem., Sect. B: Org. Chem., 2009, 105, 35–74. doi:10.1039/b822050f [Not open access]
2007
2.) “Decarboxylative Claisen rearrangements of diallyl 2-sulfonylmalonates: remarkable regioselectivity in the reaction of bifunctional substrates”, Craig, D.*; Lansdell, M.; Lewis, S. E. Tetrahedron Lett.,2007, 48, 7861–7864. doi:10.1016/j.tetlet.2007.08.130 [Not open access]
2006
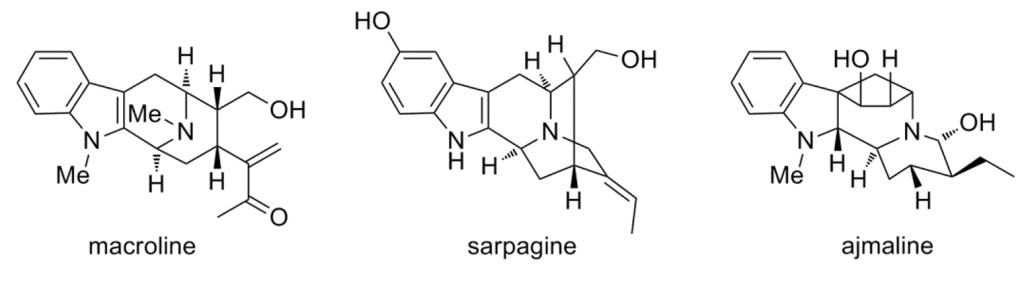
1.)“Recent advances in the chemistry of macroline, sarpagine and ajmaline-related indole alkaloids”, Lewis, S. E.* Tetrahedron, 2006, 62, 8655–8681. doi:10.1016/j.tet.2006.06.017 [Not open access]