Click on the DOIs or images to read the publications. They are all “open access” unless otherwise noted.
2026
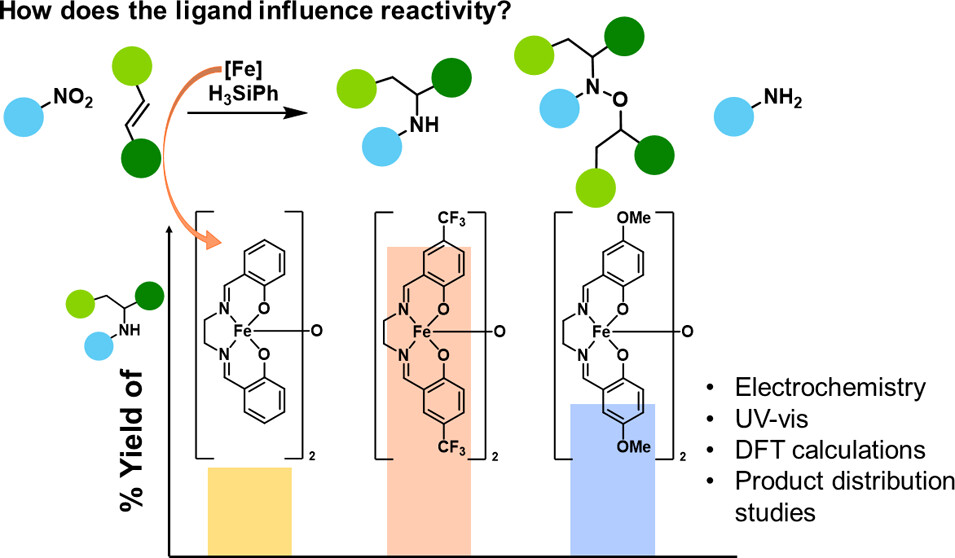
78.) “Effect of the [Fe(salen)]2‑μ-oxo catalyst electronic structure on reductive hydroamination”, Pocock, E.; Buxton, N. J.; Diefenbach, M.; Bond, A. D.; Lewis, S. E.*; Krewald, V.;* Webster, R. L.*, Inorg. Chem., 2026, 65, 999, doi:10.1021/acs.inorgchem.5c05628
2025
77.) “Synthesis and resolution of a 1,1′-biazulene analogue of BINOL”, Gee, A. P.; Gianga, T.-M.; Kociok-Köhn, G.; Pantoş, G. D.; Lewis, S. E.*, RSC Adv., 2025, 15, 14881, doi:10.1039/D5RA02520F

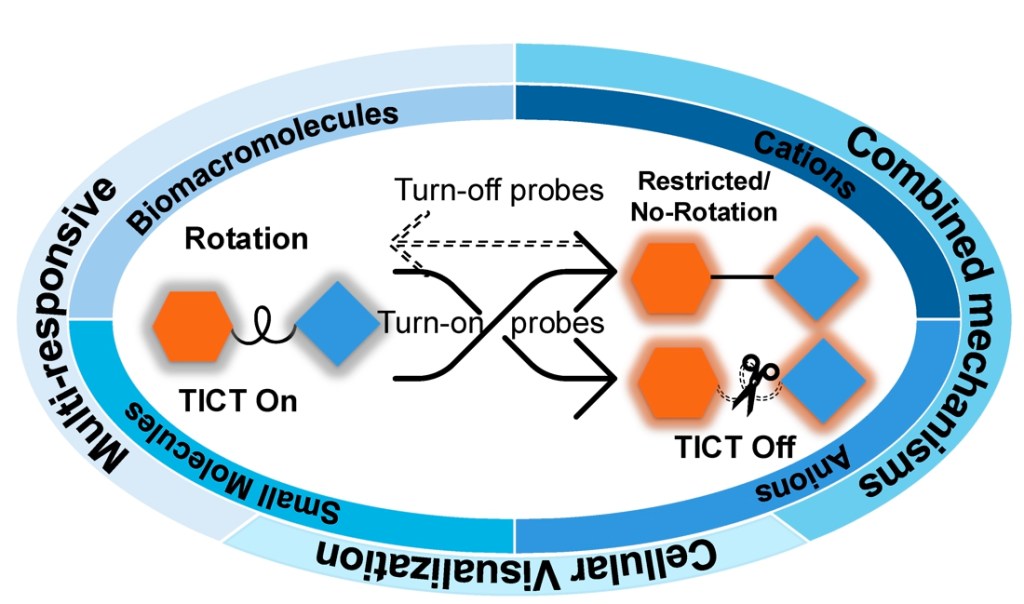
76.) “Twisted intramolecular charge transfer (TICT) based fluorescent probes and imaging agents”, Wu, Y.; Wang, H. M.; Hu, X. L.; Zang, Y.; Li, J.*; Han, H. H.*; He, X. P.*; Lewis, S. E.*; Ismail, H. M.*; James, T. D.*, Chem. Soc. Rev., 2025, 54, 12080, doi:10.1039/D3CS01118F
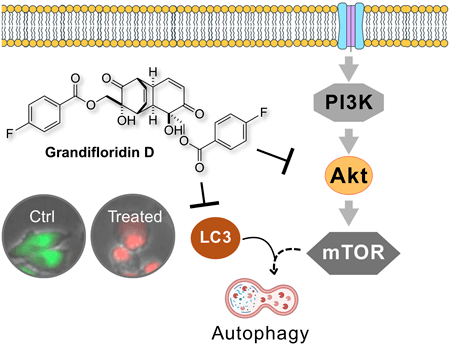
75.) “Grandifloridin D: A Potent antiausterity agent targeting pancreatic cancer cells via Akt/mTOR and autophagy inhibition”, Nguyen, H. H.; Maneenet, J. Fujii, T.; Caggiano, L.; Lewis, S. E.*; Awale, S.*, Biol. Pharm. Bull., 2025, 48, 1784, doi:10.1248/bpb.b25-00545
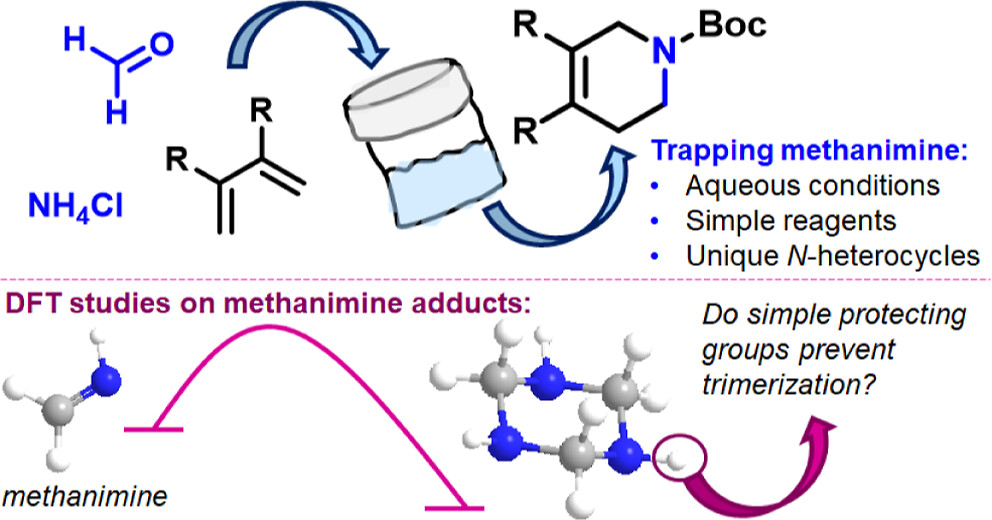
74.) “Intercepting methanimine for the synthesis of piperidine-based N-heterocycles in an aqueous medium”, Pocock, E.; Diefenbach, M.; Hood, T. M.; Nunn, M.; Krewald, V.*; Lewis, S. E.*; Webster, R. L.*, J. Org. Chem., 2025, 90, 11257, doi:10.1021/acs.joc.5c01213
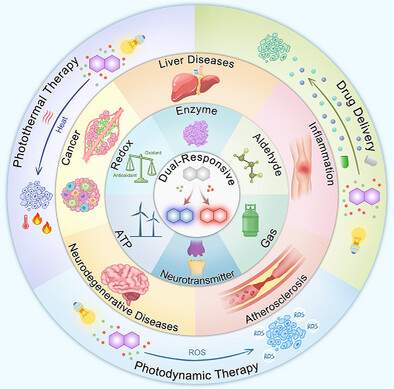
73.) “Dual-responsive fluorescent probes: Advances in biosensing, diagnosis and therapy”, Liu, J.; Wu, L.; Zhu, Z.; Yan, C.; Zhang, Y.; Yang, T.; Xu, S.; Yang, H.; Liu, S.; Tang, W.; Ma, X.; Lewis, S. E.; Wang, Q.*; James, T. D.*; Zhu, W. H.*, Adv. Funct. Mater., 2025, e15602, doi:10.1002/adfm.202515602
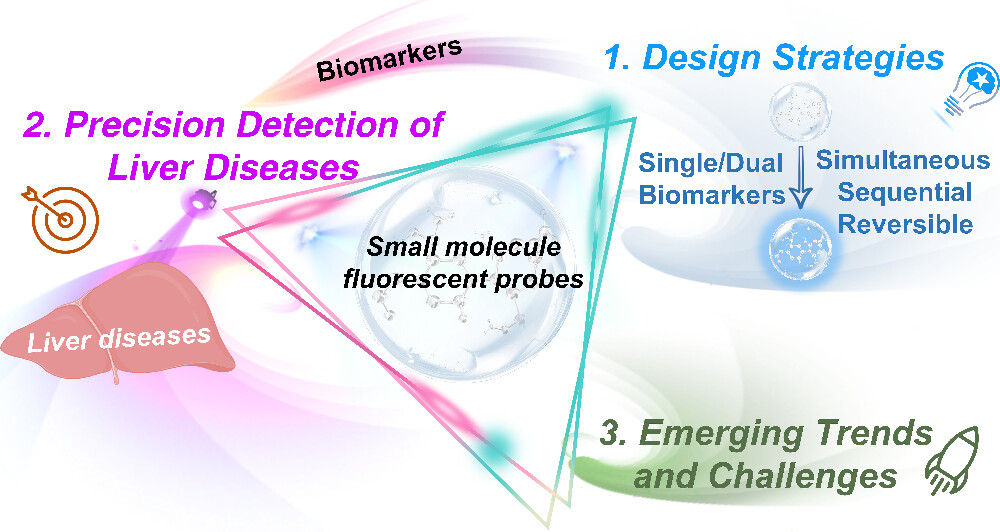
72.) “Advances in organic small molecule-based fluorescent probes for precision detection of liver diseases: A Perspective on emerging trends and challenges”, Wu, L.; Liu, Z.; Wang, K. Groleau, R. R.; Rong, X.; Liu, X.; Liu, C.; Lewis, S. E.; Zhu, B.*; James, T. D.*, J. Am. Chem. Soc., 2025, 147, 9001, doi:10.1021/jacs.4c17092
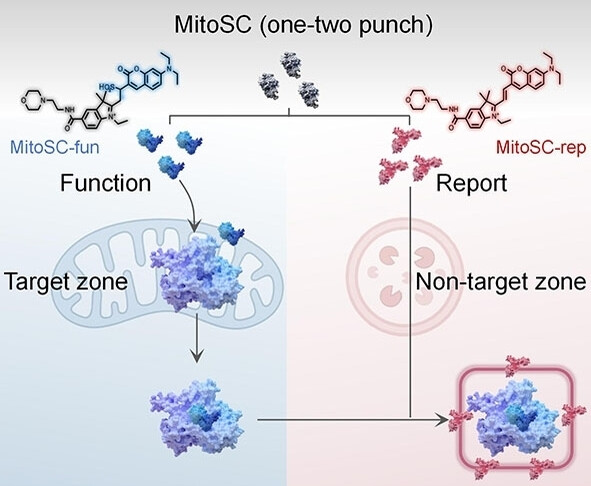
71.) “A Small-molecule drug for the self-checking of mitophagy”, Gao, Y.; Bai, Q.; Ren, Y.; Shao, X.; Zhang, M.; Wu, L.*; Lewis, S. E.; James, T. D.; Chen, X.*; Chen, Q.*, Angew. Chem. Int. Edn., 2025, 64, e202421269, doi:10.1002/anie.202421269
2024
70.) “Pyrrolylsulfonium salts: stable, accessible and versatile pseudohalides for Stille couplings”, Hann, J. L.; Lyall, C. L.; Kociok-Köhn, G.; Lewis, S. E.*, Org. Chem. Front., 2024, 11, 7011, doi:10.1039/D4QO01793E
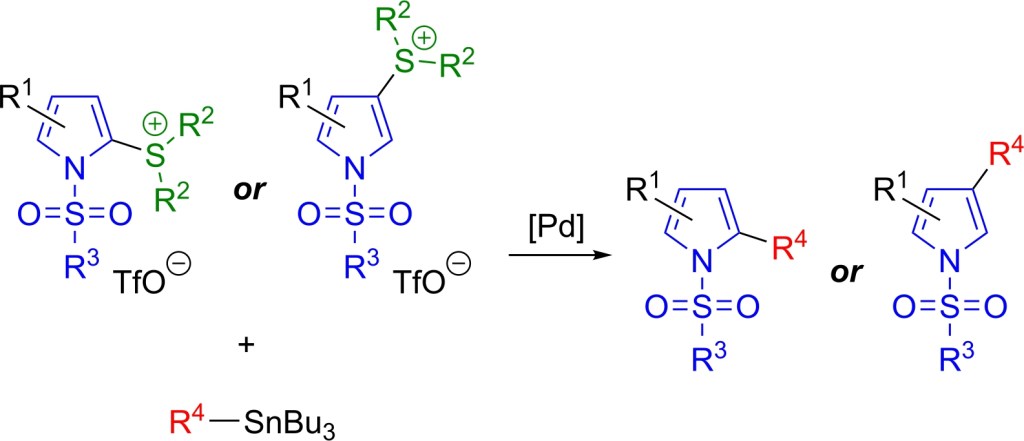
69.) “Amphiphilic azulene-based fluorescent probe for simultaneous monitoring of fluctuations in carboxylesterase activity in diverse biological samples from a single organism”, Cui, Z.; Wang, Y.; Wang, G.; Feng, B.; Lewis, S. E.; Wang, K.; Jiang, K.; James, T. D.*; Zhang, H.*, Anal. Chem., 2024, 96, 19732, doi:10.1021/acs.analchem.4c04926
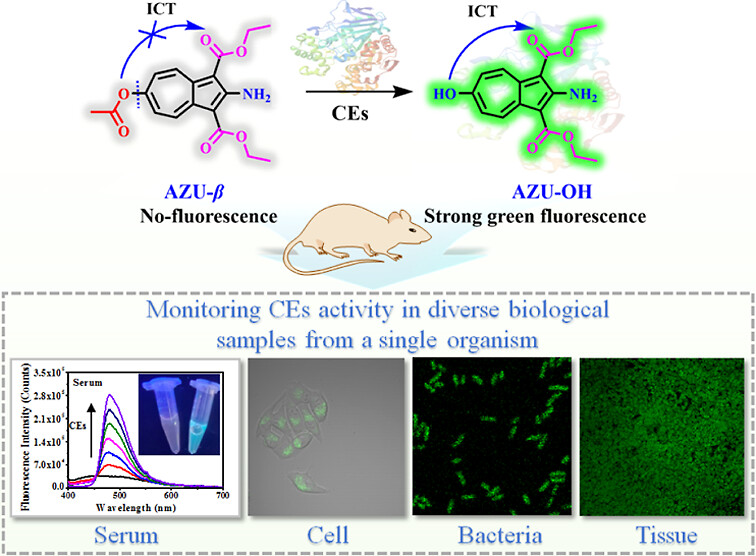
68.) “Carbon–carbon bond formation using aromatics from biomass”, Dunås, P.; Patterson, A. J.; Lewis, S. E.*; Kann, N.*, Chem. Commun., 2024, 60, 14885, doi:10.1039/D4CC05664G
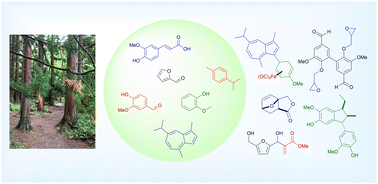
67.) “Unusual regio- and chemoselectivity in oxidation of pyrroles and indoles enabled by a thianthrenium salt intermediate”, Hann, J. L.; Lyall, C. L.; Kociok-Köhn, G.; Faverio, C.; Pantoş, G. D.*; Lewis, S. E.*, Angew. Chem. Int. Edn., 2024, 63, e202405057, doi:10.1002/anie.202405057
66.) “Fluorescent probes for disease diagnosis”, Wang, X., Ding, Q., Groleau, R. R., Wu, L., Mao, Y., Che, F., Kotova, O., Scanlan, E. M., Lewis, S. E.*, Li, P.*, Tang, B.*, James, T. D.*; Gunnlaugsson, T.*, Chem. Rev., 2024, 124, 7106, doi:10.1021/acs.chemrev.3c00776
65.) “Fluorescent small molecule donors”, Chen, G.*; Yu, J.; Wu, L.; Ji, X.; Xu, J.; Wang, C.; Ma, S.; Miao, Q.; Wang, L.; Wang, C.; Lewis, S. E.; Yue, Y.*; Sun, Z.*; Liu, Y.*; Tang, B.*; James, T. D.*, Chem. Soc. Rev., 2024, 53, 6345, doi:10.1039/D3CS00124E
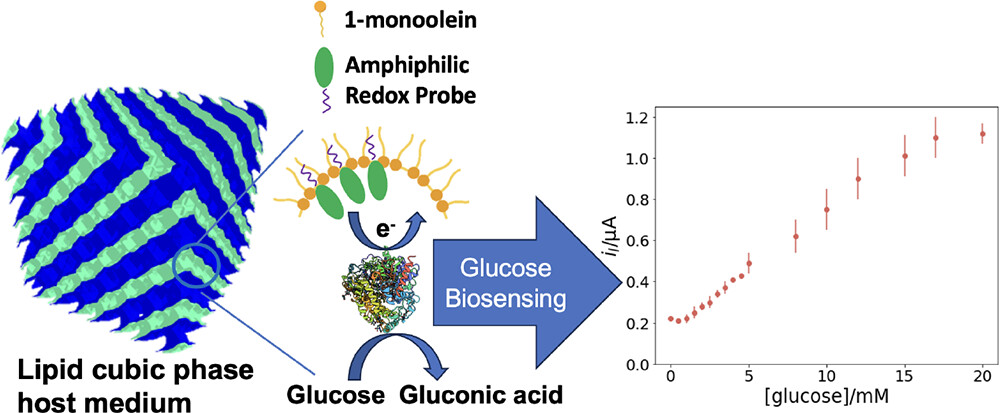
64.) “Development of redox-active lyotropic lipid cubic phases for biosensing platforms”, Liu, W.; Lewis, S. E.; Di Lorenzo, M.; Squires, A. M.*; Langmuir, 2024, 40, 170, doi:10.1021/acs.langmuir.3c02307
2023
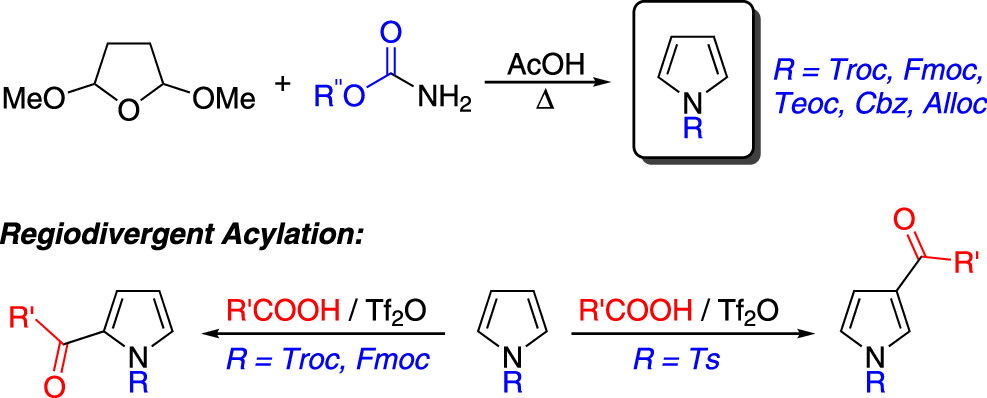
63.) “Synthesis of N-alkoxycarbonyl pyrroles from O-substituted carbamates: A synthetically enabling pyrrole protection strategy”, Hann, J. L.; Lyall, C. L. , Kociok-Köhn, G.; Lewis, S. E.*, J. Org. Chem., 2023, 88, 13584, doi:10.1021/acs.joc.3c01257
62.) “Photochemical fingerprinting is a sensitive probe for the detection of synthetic cannabinoid receptor agonists; Toward robust point-of-care detection”, Andrews, R.; May, B.; Hernández, F. J.; Townsend, P.; Cozier, G.; Sutcliffe, O. B.; Haines, T. S. F.; Freeman, T.; Scott, J.; Husbands, S.; Blagbrough, I.; Bowman, R.; Lewis, S. E.; Grayson, M.; Crespo-Otero, R.*; Carbery, D.*; Pudney, C.*, Anal. Chem., 2023, 95, 703, doi:10.1021/acs.analchem.2c02529
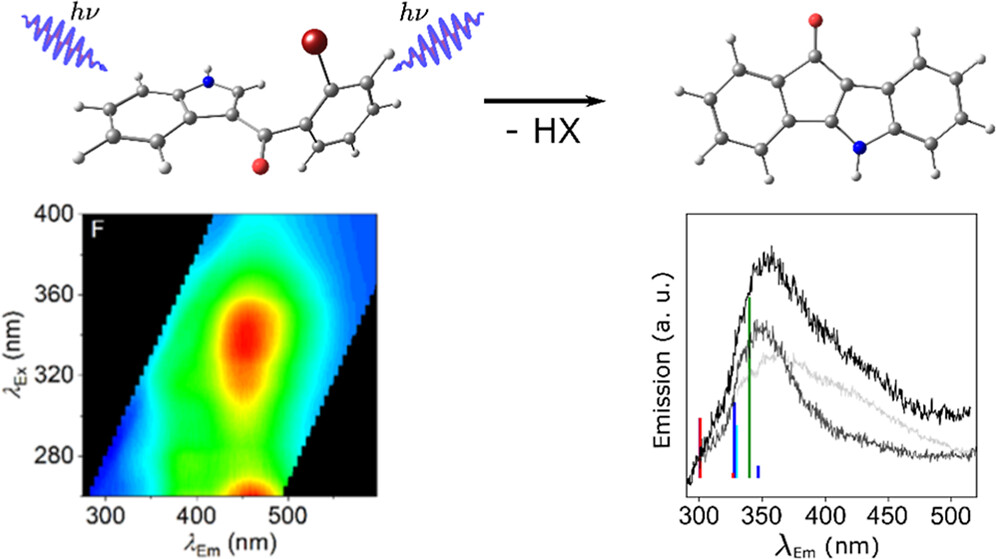
61.) “Persistent azulene α-carbocations: synthesis from aldehydes, spectroscopic and crystallographic properties”, Harabajiu, C.; Hann, J. L; Murfin, L. C., Kociok-Köhn, G.; Lewis, S. E.*, Org. Biomol. Chem., 2023, 21, 858 doi:10.1039/D2OB01695H

2022
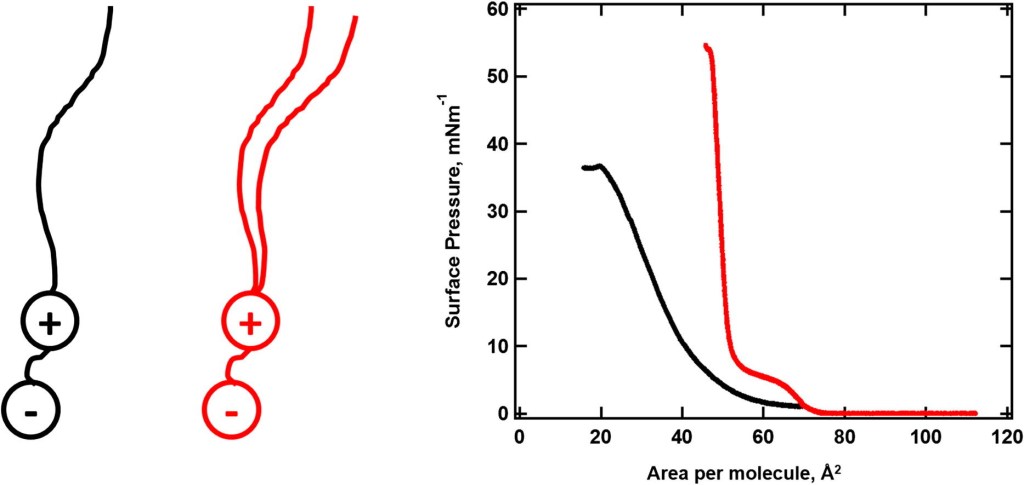
60.) “Structural investigation of sulfobetaines and phospholipid monolayers at the air–water interface”, Elstone, N.*; Arnold, T.; Skoda, M. W. A.; Lewis, S. E.; Li, P.; Hazell, G.; Edler, K. J.*, Phys. Chem. Chem. Phys., 2022, 24, 22679, doi:10.1039/d2cp02695c
2021
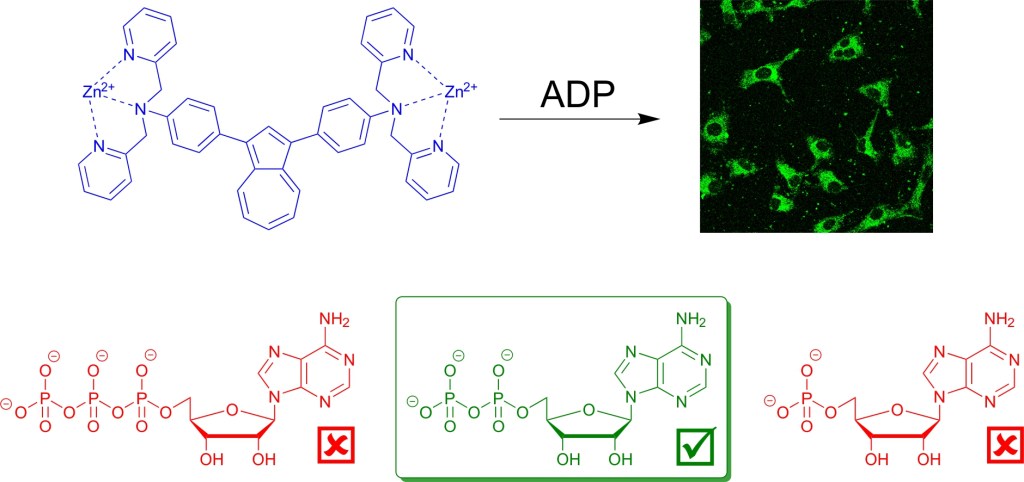
59.) “Azulene-based fluorescent chemosensor for adenosine diphosphate”, López-Alled, C. M.; Park, S. J.; Lee, D. J.; Murfin, L. C.; Kociok-Köhn, G.; Hann, J. L.; Wenk, J.*; James, T. D.*; Kim, H. M.*; Lewis, S. E.*, Chem. Commun., 2021, 57, 10608, doi:10.1039/d1cc04122c
58.) “Palladium-catalyzed stereoselective domino arylation–acylation: an entry to chiral tetrahydrofluorenone scaffolds”, Dunås, P.; Paterson, A. J.; Kociok-Köhn, G.; Rahm, M.; Lewis, S. E.*; Kann, N.*, Chem. Commun., 2021, 57, 6518, doi:10.1039/d1cc02160e

57.) “Azulene – A bright core for sensing and imaging”, Murfin, L. C.*; Lewis, S. E., Molecules, 2021, 26, 353, doi:10.3390/molecules26020353
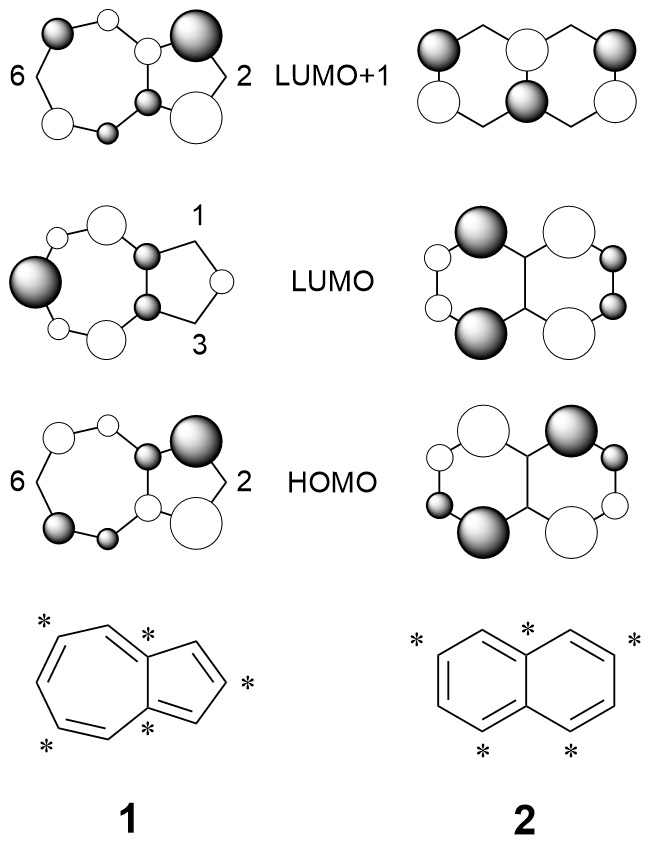
56.) “C4-aldehyde of guaiazulene: synthesis and derivatisation”, Williams, G. E.; Kociok-Köhn, G.; James, T. D.; Lewis, S. E.*, Org. Biomol. Chem., 2021, 19, 2502, doi:10.1039/d0ob02567d
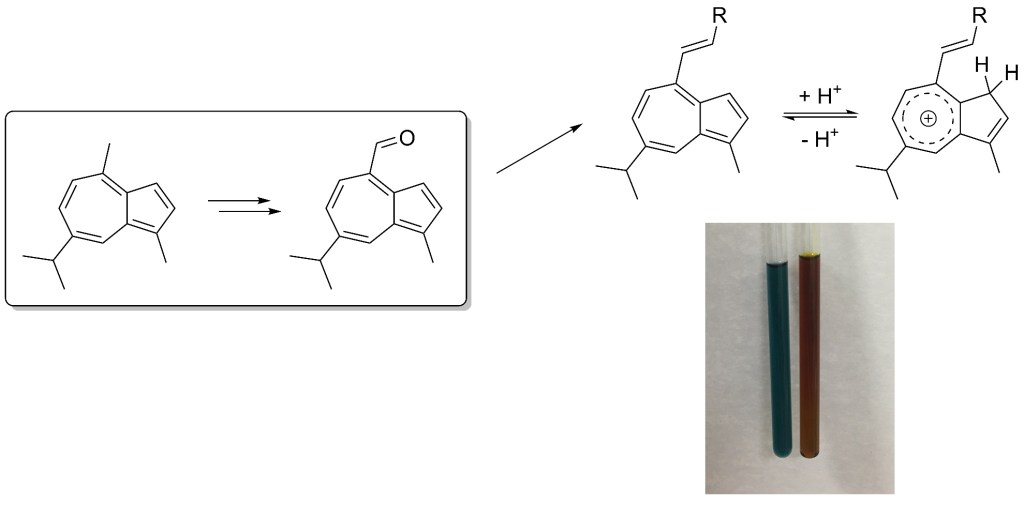
55.) “Fluorescent small organic probes for biosensing”, Tian, X.; Murfin, L. C.; Wu, L.*; Lewis, S. E.*; James, T. D.*, Chem. Sci., 2021, 12, 3406, doi:10.1039/D0SC06928K
2020
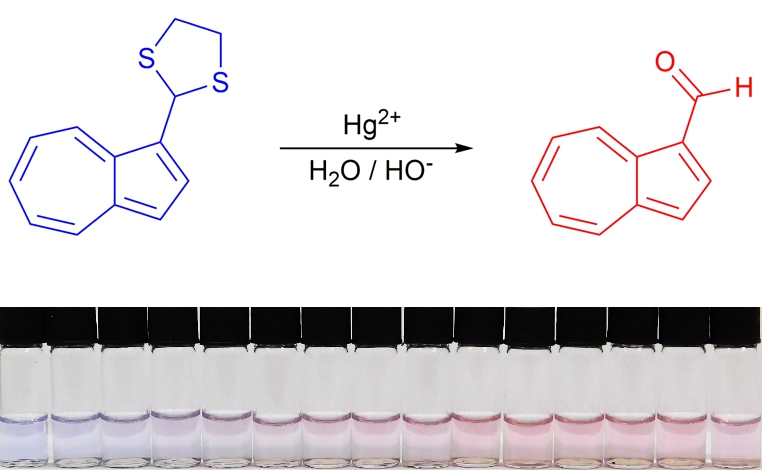
54.) “Colorimetric detection of Hg2+ with an azulene-containing chemodosimeter via dithioacetal hydrolysis”, López-Alled, C. M.; Murfin, L. C.; Kociok-Köhn, G.; James, T. D.*; Wenk, J.*; Lewis, S. E.*, Analyst, 2020, 145, 6262, doi:10.1039/d0an01404d
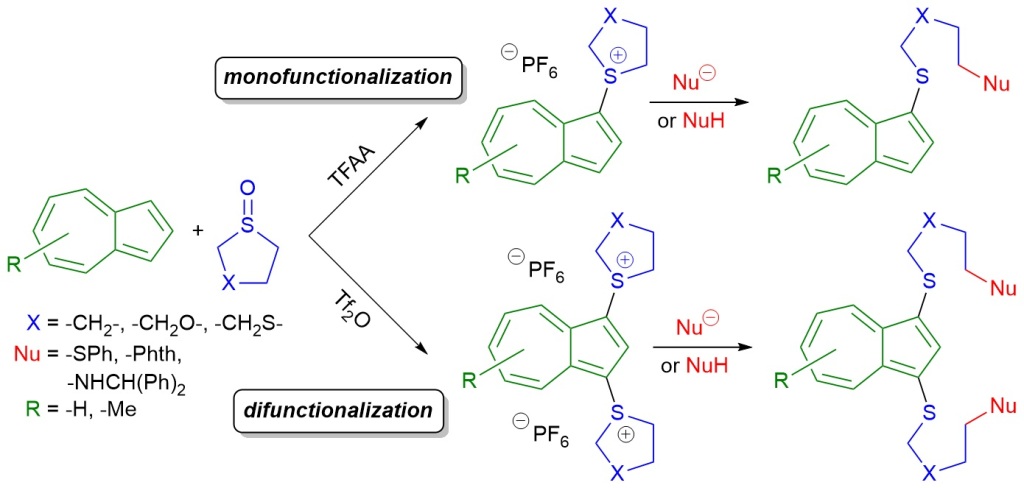
53.) “Azulenesulfonium and azulenebis(sulfonium) salts: Formation by interrupted Pummerer reaction and subsequent derivatisation by nucleophiles”, López-Alled, C. M.; Martin, F. J. O.; Chen, K.-Y.; Kociok-Köhn, G.; James, T. D.; Wenk, J.; Lewis, S. E.*, Tetrahedron, 2020, 76, 131700, doi:10.1016/j.tet.2020.131700
52.) “Azulene functionalization by iron-mediated addition to a cyclohexadiene scaffold”, Dunås, P.; Murfin, L. C.; Nilsson, O. J.; Jame, N.; Lewis, S. E.*; Kann, N.*, J. Org. Chem., 2020, 85, 13453, doi:10.1021/acs.joc.0c01412
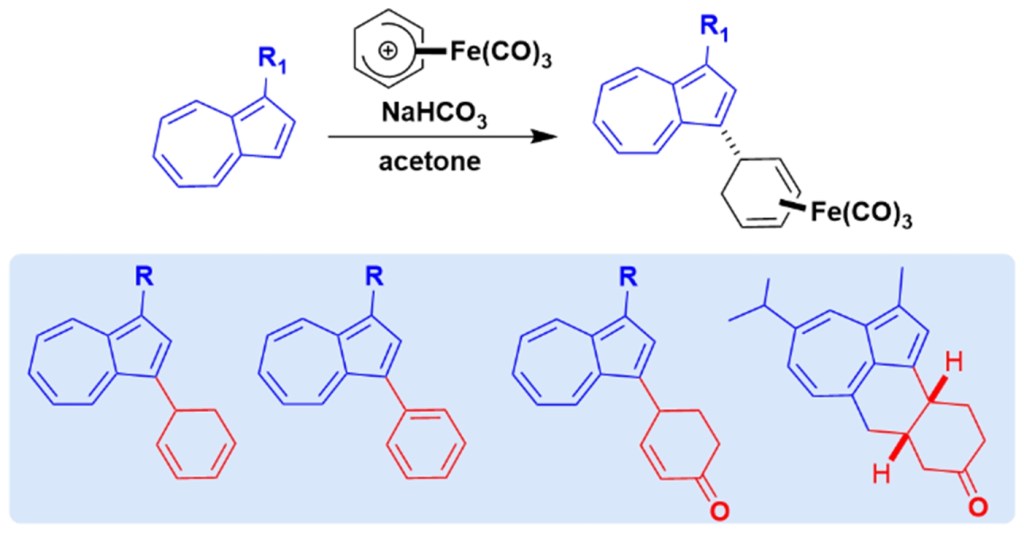
51.) “Palladium catalyzed stereoselective arylation of biocatalytically derived cyclic 1,3-dienes: Chirality transfer via a Heck-type mechanism”, Paterson, A. J.; Dunås, P.; Rahm, M.; Norrby, P.-O.; Kociok-Köhn, G.; Lewis, S. E.*; Kann, N.*, Org. Lett., 2020, 22, 2464, doi:10.1021/acs.orglett.0c00708
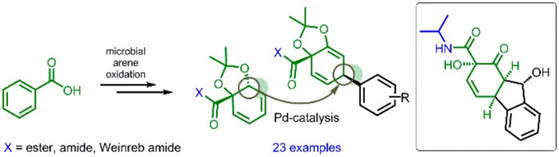
50.) “(Fluoro)quinolones and quinolone resistance genes in the aquatic environment: A river catchment perspective”, Castrignanò, E.; Kannan, A. M.; Proctor, K.; Petrie, B.; Hodgen, S.; Feil, E. J.; Lewis, S. E.; Lopardo, L.; Camacho-Muñoz, D.; Rice, J.; Cartwright, N.; Barden, R.; Kasprzyk-Hordern, B.*, Water Res., 2020, 182, 116015, doi:10.1016/j.watres.2020.116015
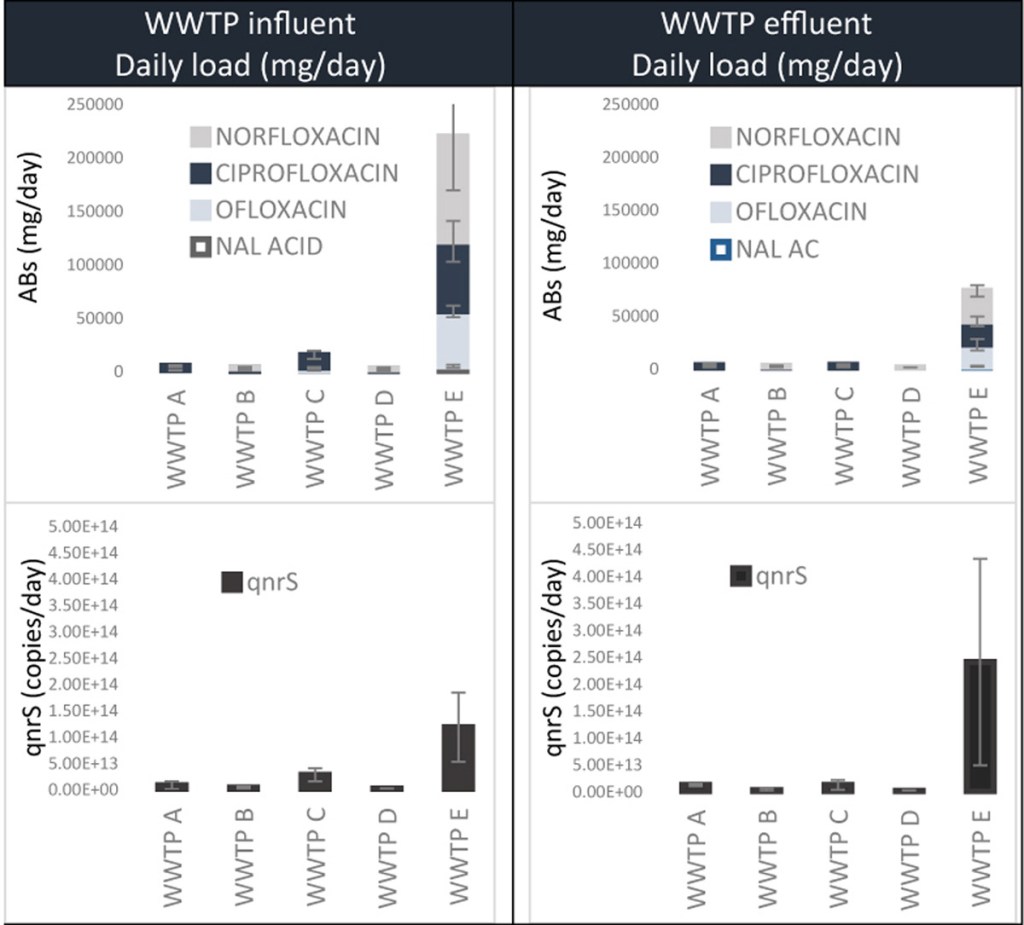
49.) “A Colorimetric chemosensor based on a Nozoe azulene that detects fluoride in aqueous/alcoholic media”, Murfin, L. C.; Chiang, K. X. C.; Lyall, C. L.; Williams, G. T.; Wenk, H.*; Jenkins, T.*; James, T. D.*; Lewis, S. E.*, Front. Chem., 2020, doi: 10.3389/fchem.2020.00010

48.) “A simple, azulene-based colorimetric probe for the detection of nitrite in water”, Murfin, L. C.; López-Alled, C. M.; Sedgwick, A. C.; Wenk, J.*; James, T. D.*; Lewis, S. E.*, Front. Chem. Sci. Eng., 2020, doi:10.1007/s11705-019-1790-7

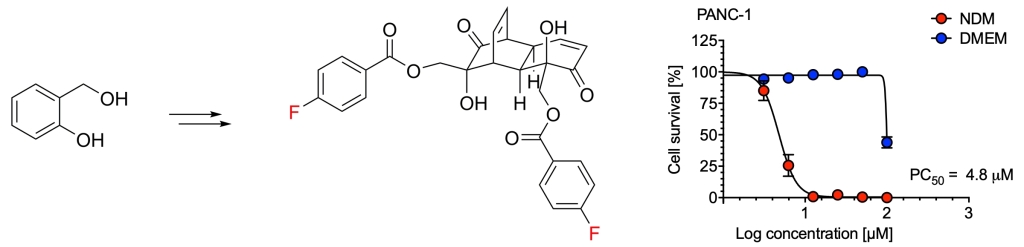
47.) “Sidechain Diversification of Grandifloracin Allows Identification of Analogues with Enhanced Anti‐Austerity Activity against Human PANC‐1 Pancreatic Cancer Cells”, Alexander, B. E.; Sun, S.; Palframan, M. J.; Kociok‐Köhn, G.; Dibwe, D. F.; Watanabe, S.; Caggiano, L.*; Awale, S.*; Lewis, S. E.*, ChemMedChem, 2020, 15, 125, doi:10.1002/cmdc.201900549
2019
46.) “Azulene-Derived Fluorescent Probe for Bioimaging: Detection of Reactive Oxygen and Nitrogen Species by Two-Photon Microscopy”, Murfin, L. C.; Weber, M.; Park, S. J.; Kim, W. T.; Lopez-Alled, C. M.; McMullin, C. L.; Pradaux-Caggiano, F.; Lyall, C. L.; Kociok-Köhn, G.; Wenk, J. H.; Bull, S. D.; Yoon, J.; Kim, H. M.*; James, T. D.*; Lewis, S. E.*, J. Am. Chem. Soc. 2019, 141, 19389, doi: 10.1021/jacs.9b09813

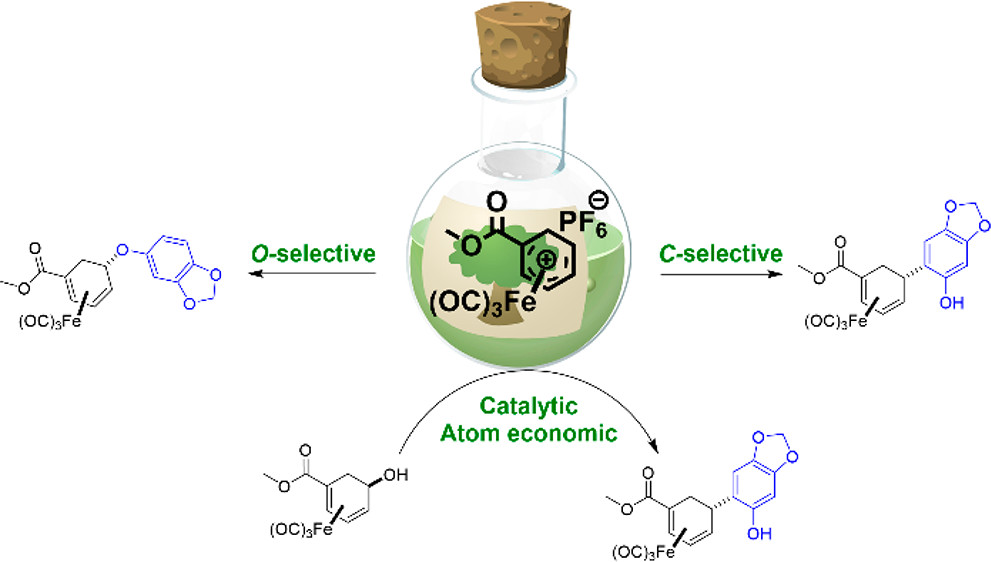
45.) “Selective Iron-Mediated C- and O-Addition of Phenolic Nucleophiles to a Cyclohexadiene Scaffold Using Renewable Precursors”, Dunås, P.; Paterson, A. J.; Kociok-Köhn, G.; Lewis, S. E.*; Kann, N.*, ACS Sustainable Chem. Eng., 2019, 7, 7155, doi:10.1021/acssuschemeng.9b00127
44.) “Partial Cation Substitution Reduces Iodide Ion Transport in Lead Iodide Perovskite Solar Cells”, Ferdani, D.; Pering, S.; Ghosh, D.; Kubiak, P.; Walker, A.; Lewis, S. E.; Johnson, A. L.; Baker, P. J.; Islam, M. S.; Cameron, P. J., Energy Environ. Sci., 2019, 12, 2264, doi: 10.1039/c9ee00476a

43.) “Azulenes with aryl substituents bearing pentafluorosulfanyl groups: synthesis, spectroscopic and halochromic properties”, Webster, S. J.; López-Alled, C. M.; Liang, X.; McMullin, C. L.; Kociok-Köhn, G.; Lyall, C. L.; James, T. D.; Wenk, J.; Cameron, P. J.; Lewis, S. E.*, New J. Chem., 2019, 43, 992, doi:10.1039/C8NJ05520C

2018
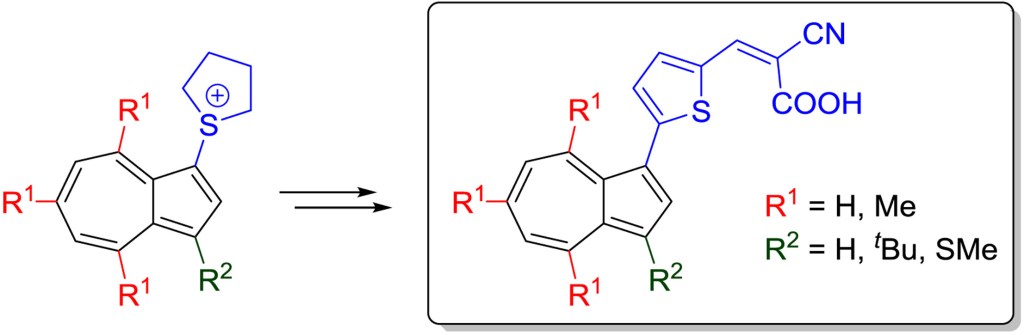
42.) “Azulene–Thiophene–Cyanoacrylic acid dyes with donor-π-acceptor structures. Synthesis, characterisation and evaluation in dye-sensitized solar cells”, Cowper, P*; Pockett, A.; Kociok-Köhn, G.; Cameron, P. J.; Lewis, S. E*, Tetrahedron, 2018, 74, 2775, doi:10.1016/j.tet.2018.04.043
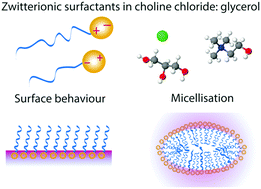
41.) “Self-assembly and surface behaviour of pure and mixed zwitterionic amphiphiles in a deep eutectic solvent”, Sanchez-Fernandez, A.; Moody, G. L.; Murfin, L. C.; Arnold,T.; Jackson, A. J.; King, S. M.; Lewis, S. E.; Edler, K. J., Soft Matter, 2018, 14, 5525-5536. doi:10.1039/c8sm00755a
2017
40.) “Enantioselective transformation of fluoxetine in water and its ecotoxicological relevance”, Andrés-Costa, M. J.; Proctor, K.; Sabatini, M. T.; Gee, A. P.; Lewis, S. E.; Pico, Y.; Kasprzyk-Hordern, B., Sci. Rep., 2017, 7, 15777. doi:10.1038/s41598-017-15585-1

39.) “Azetidinium lead iodide for perovskite solar cells”, Pering, S. R.; Deng, W.; Troughton, J. R.; Kubiak, P. S.; Ghosh, D.; Niemann, R. G.; Brivio, F.; Jeffrey, F. E.; Walker, A. B.; Islam, M. S.; Watson, T. M.; Raithby, P. R.; Johnson, A. L.; Lewis, S. E.; Cameron; P. J.*, J. Mater. Chem. A, 2017,5, 20658-20665. doi:10.1039/C7TA07545F
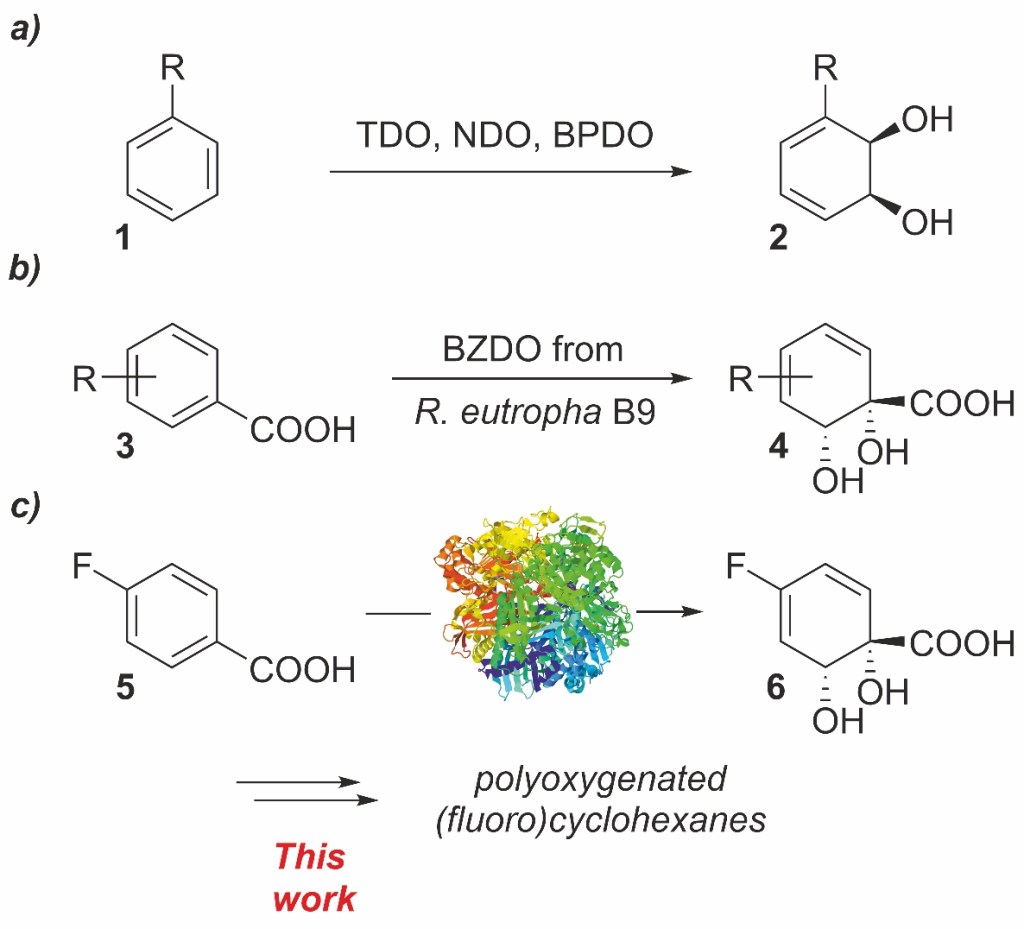
38.) “Biocatalytic dearomatisation of para-fluorobenzoic acid – Access to versatile homochiral building blocks with quaternary centres”, Nash, T. J.; Wharry, S.; Moody, T. S.*; Lewis, S. E.*, Chimica Oggi, 2017, 35, 90, http://www.teknoscienze.com/tks_article/biocatalytic-dearomatisation-of-para-fluorobenzoic-acid-access-to-versatile-homochiral-building-blocks-with-quaternary-centres/
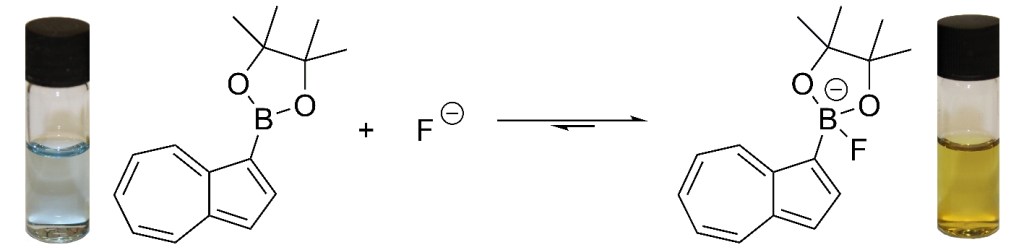
37.) “Azulene–boronate esters: colorimetric indicators for fluoride in drinking water”, López-Alled, C. M.; Sanchez-Fernandez, A.; Edler, K. J.; Sedgwick, A. C.; Bull, S. D.; McMullin, C. L.; Kociok-Köhn, G.; James, T. D.*; Wenk, J.*; Lewis, S. E.*, Chem. Commun., 2017, 53, 12580, doi:10.1039/c7cc07416f
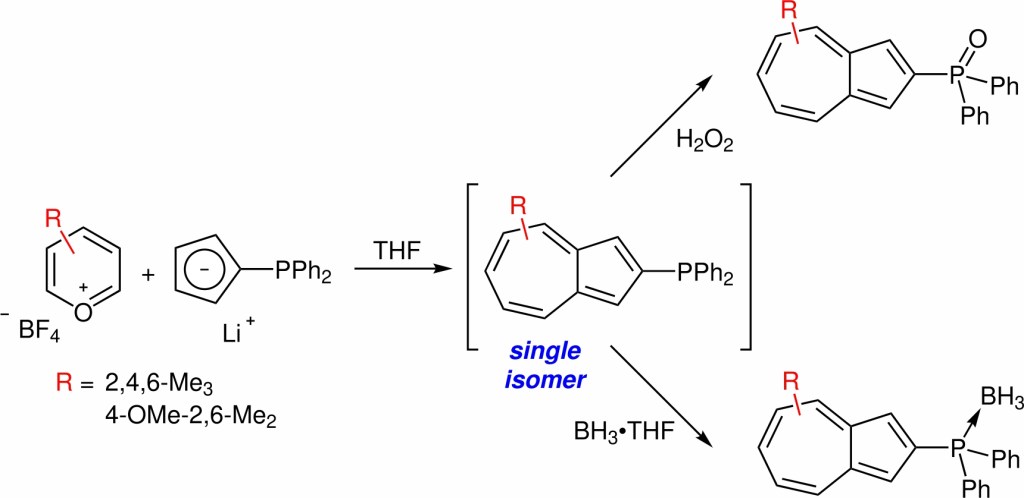
36.) “Phosphorus-substituted azulenes accessed via direct Hafner reaction of a phosphino cyclopentadienide”, Gee, A. P.; Cosham, S. D.; Johnson, A. L.; Lewis, S. E.* Synlett, 2017, 973, doi:10.1055/s-0036-1589936
2016
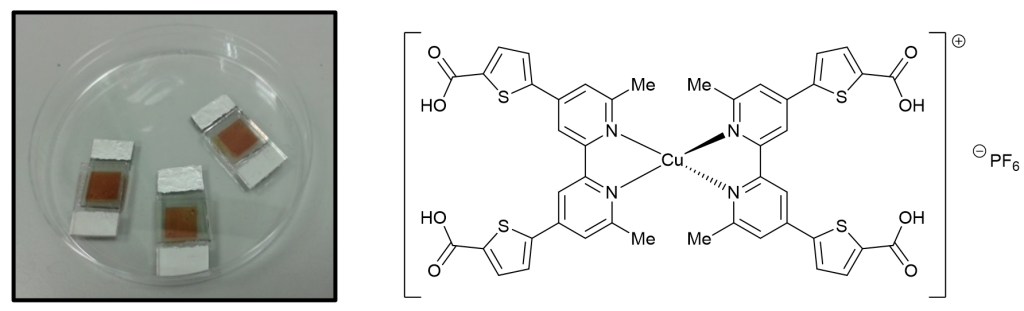
35.) “What difference does a thiophene make? Evaluation of a 4,4’-bis(thiophene) functionalised 2,2’-bipyridyl copper(I) complex in a dye-sensitized solar cell”, Wills, K. A.; Mandujano-Ramírez, H. A.; Merino, G.; Oskam, G.; Cowper, P.; Jones, M. D.*; Cameron, P. J.*; Lewis, S. E.* Dyes and Pigments, 2016, 134, 419, doi:10.1016/j.dyepig.2016.07.023
34.) “Asymmetric Dearomatization Under Enzymatic Conditions”, Lewis, S. E. in Asymmetric Dearomatization Reactions (You, S.-L. ed.) Wiley-VCH, 2016, pp 279–346. doi:10.1002/9783527698479.ch12
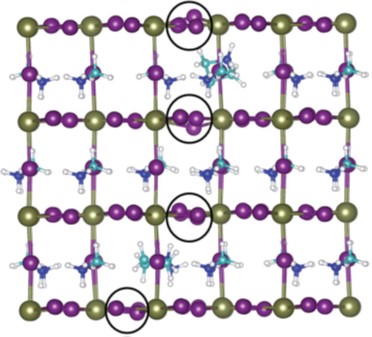
33.) “Langmuir monolayers composed of single and double tail sulfobetaine lipids”, Hazell, G.; Gee, A. P.; Arnold, T.; Edler, K. J.*; Lewis, S. E. J. Colloid Interface Sci., 2016, 474, 190, doi:10.1016/j.jcis.2016.04.020
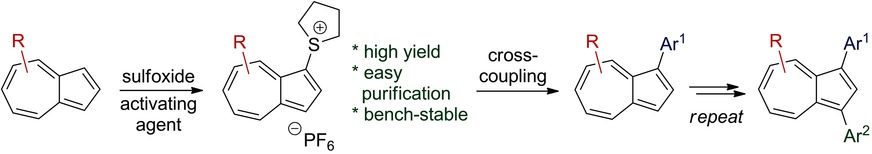
32.) “Azulenesulfonium Salts: Accessible, Stable and Versatile Reagents for Cross-Coupling”, Cowper, P.*; Jin, Y.; Turton, M. D.; Kociok-Köhn, G.; Lewis, S. E.* Angew. Chem. Int. Edn., 2016, 55, 2564, doi:10.1002/anie.201510666
2015
31.) “Biotransformations of Arenes: An Overview”, Lewis, S. E. in Arene Chemistry: Reaction Mechanisms and Methods for Aromatic Compounds (Mortier, J. ed.) Wiley-VCH, 2015, pp 915–937. doi:10.1002/9781118754887.ch32
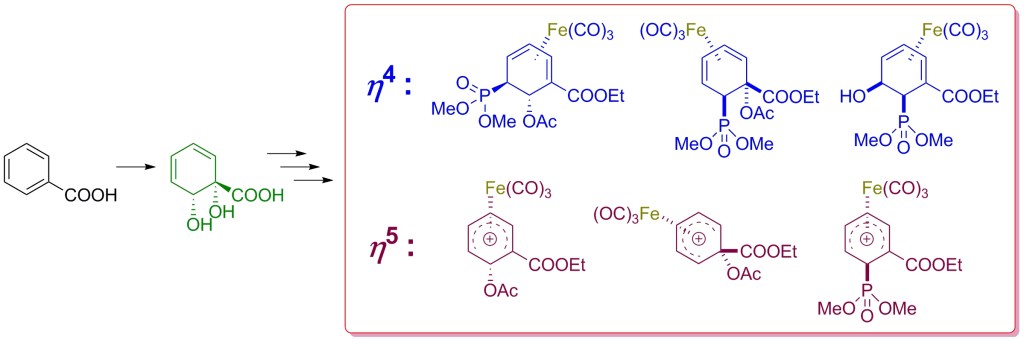
30.) “Tricarbonyliron(0) complexes of bio-derived η4 cyclohexadiene ligands: An approach to analogues of oseltamivir”, ten Broeke, M.; Ali Khan, M.; Kociok-Köhn, G.; Kann, N.; Lewis, S. E.* J. Organomet. Chem., 2015, 799-800, 19, doi:10.1016/j.jorganchem.2015.09.005
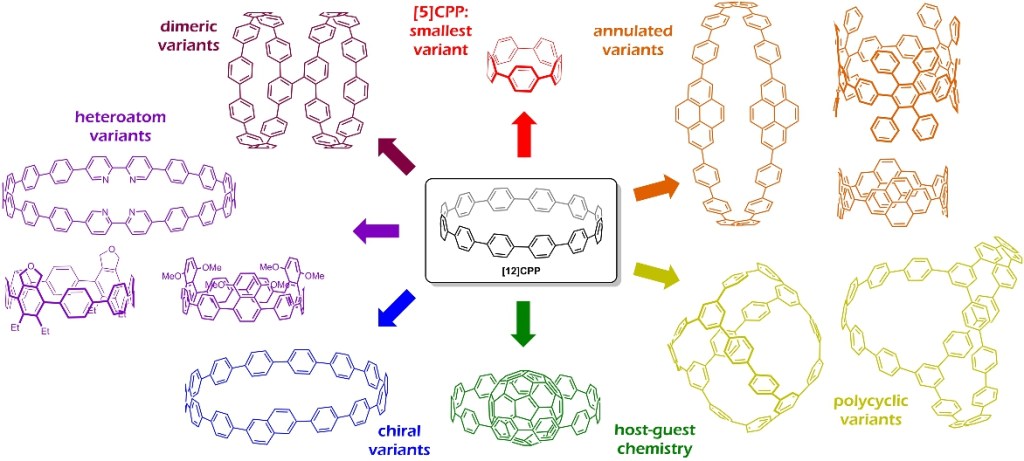
29.) “Cycloparaphenylenes and related nanohoops”, Lewis, S. E.* Chem. Soc. Rev., 2015, 44, 2221–2304. doi:10.1039/c4cs00366g
2014
28.) “Direct core functionalisation of naphthalenediimides by iridium catalysed C–H borylation”, Lyall, C. L.; Shotton, C. C.; Pérez-Salvia, M.; Pantoş, G. D.*; Lewis, S. E.* Chem. Commun., 2014, 50, 13837, doi:10.1039/c4cc06522k
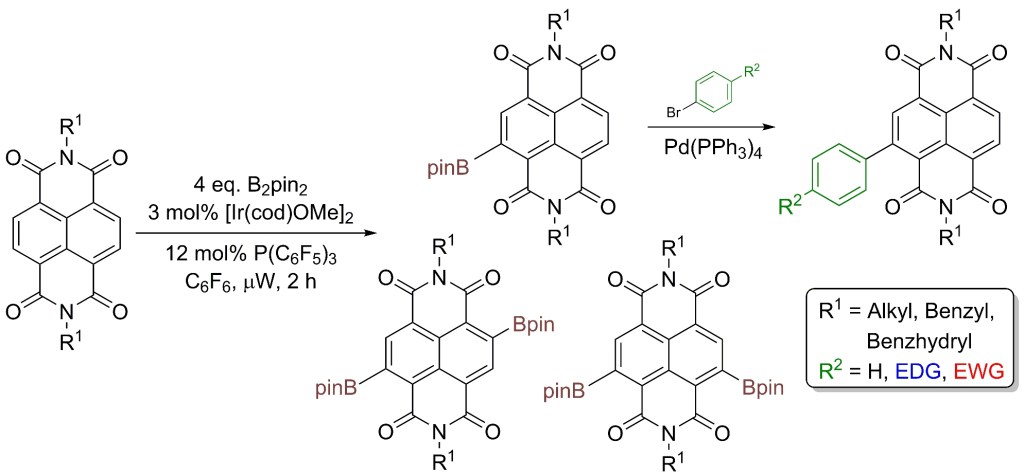
27.) “C–H functionalization of sp3 centers with aluminum: a computational and mechanistic study of the Baddeley reaction of decalin”, Lyall,C. L.; Sato, M.; Uosis-Martin, M.; Asghar, S. F.; Jones, M. D.; Williams, I. H.*; Lewis, S. E.* J. Am. Chem. Soc., 2014, 136, 13745, doi:10.1021/ja5062246
26.) “A new assay for rhamnolipid detection—important virulence factors of Pseudomonas aeruginosa”, Laabei, M.; Jamieson, W. D.; Lewis, S. E.; Diggle, S. P.; Jenkins, A. T. A.* Appl. Microbiol. Biotech., 2014, 98, 7199, doi:10.1007/s00253-014-5904-3
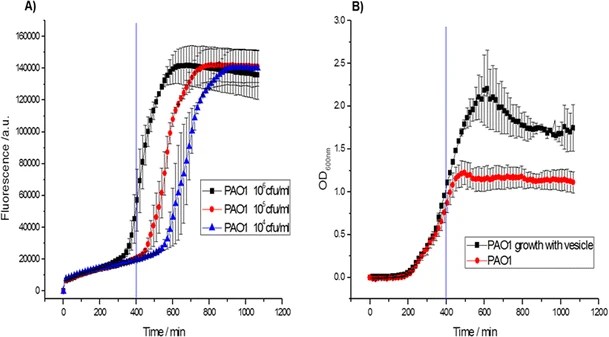
25.) “The Enone Motif of (+)-Grandifloracin is Not Essential for “Anti-Austerity” Antiproliferative Activity”, Ali Khan, M.; Wood, Pauline J.; Lamb-Guhren, N. M.; Caggiano, L.; Kociok-Köhn, G.; Tosh, D.; Lewis, S. E.* Bioorg. Med. Chem. Lett., 2014, 24, 2815, doi:10.1016/j.bmcl.2014.04.111
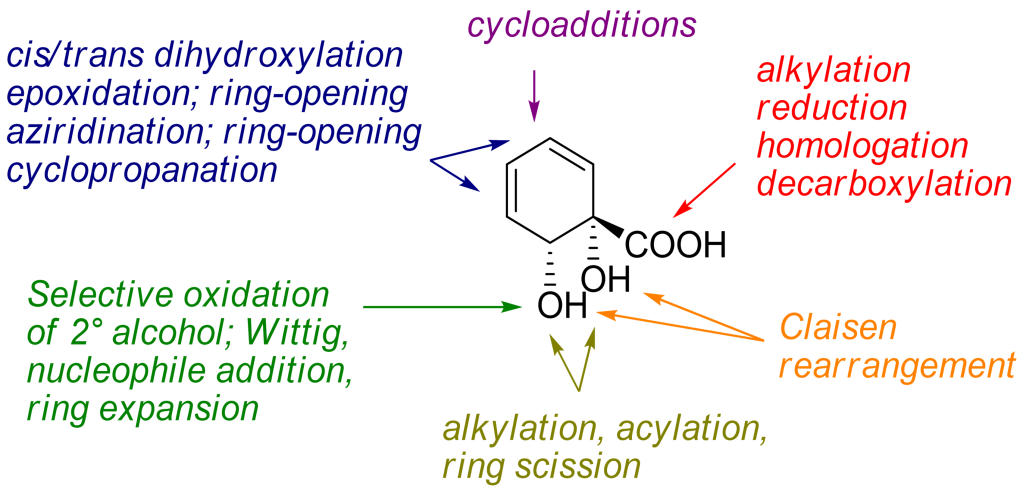
24.) “Applications of biocatalytic arene ipso,orthocis-dihydroxylation in synthesis”, Lewis, S. E.* Chem. Commun., 2014, 50, 2821, doi:10.1039/c3cc49694e
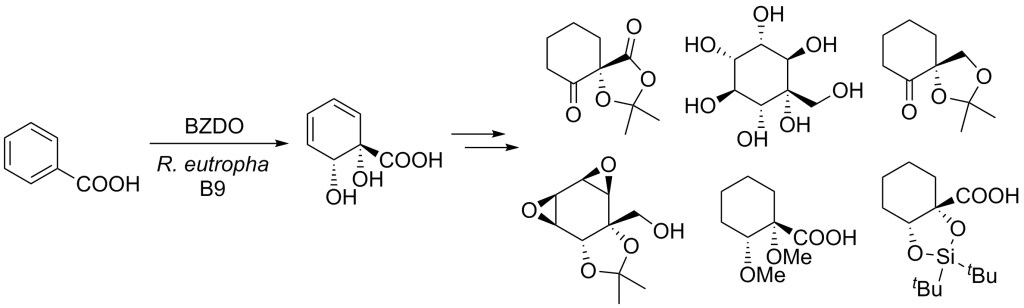
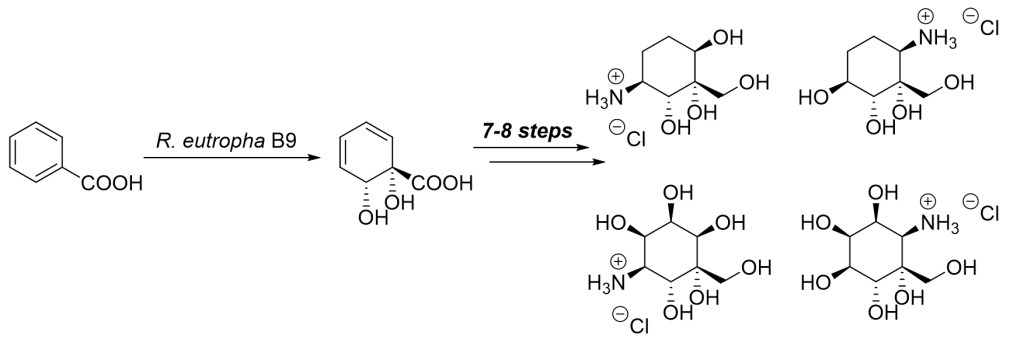
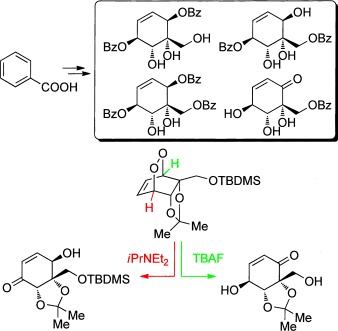
23.) “Benzoate dioxygenase from Ralstonia eutropha B9 – unusual regiochemistry of dihydroxylation permits rapid access to novel chirons”, Griffen, J. A.; Kenwright, S. J.; Abou-Shehada, S.; Wharry, S.; Moody, T. S.*; Lewis, S. E.* Org. Chem. Front., 2014, 1, 79, doi:10.1039/c3qo00057e
2013
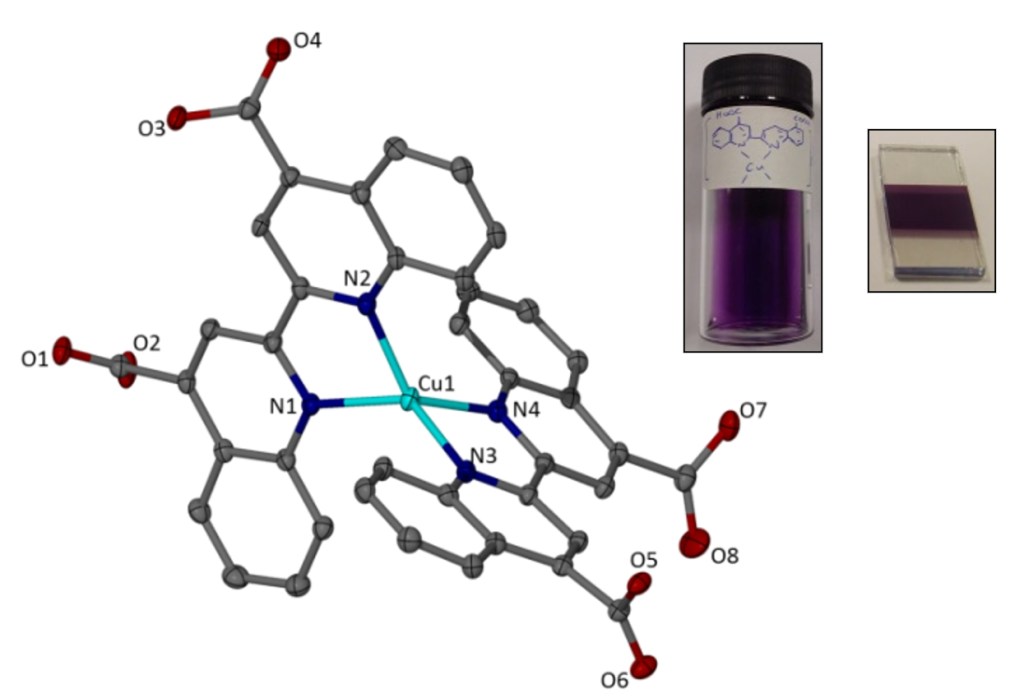
22.) “Investigation of a copper(I) biquinoline complex for application in dye-sensitized solar cells”, Wills, K. A.; Mandujano-Ramírez, H. J.; Merino, G.; Mattia, D.; Hewat, T.; Robertson, N.; Oskam, G.; Jones, M. D.; Lewis, S. E.; Cameron, P. J.*, RSC Advances, 2013, 3, 23361, doi:10.1039/c3ra44936j
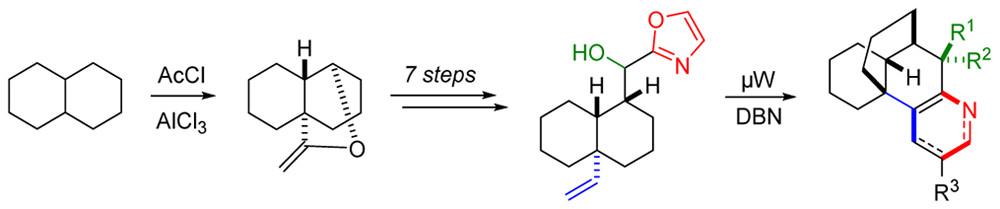
21.) “A Model System for the Synthesis of Complanadine Alkaloids by ‘Diverted Kondrat’eva’ Oxazole–Olefin Cycloaddition”, Uosis-Martin, M.; Pantoş, G. D.; Mahon, M. F.; Lewis, S. E.* J. Org. Chem., 2013, 78, 6253, doi:10.1021/jo401014n
20.) “New Aminocyclitols with Quaternary Stereocentres via Acylnitroso Cycloaddition with an ipso,ortho-Arene Dihydrodiol”, Griffen, J. A.; White, J. C.; Kociok-Köhn, G.; Lloyd, M. D.; Wells, A.; Arnot, T. C.; Lewis, S. E.* Tetrahedron, 2013, 69, 5989, doi:10.1016/j.tet.2013.04.033
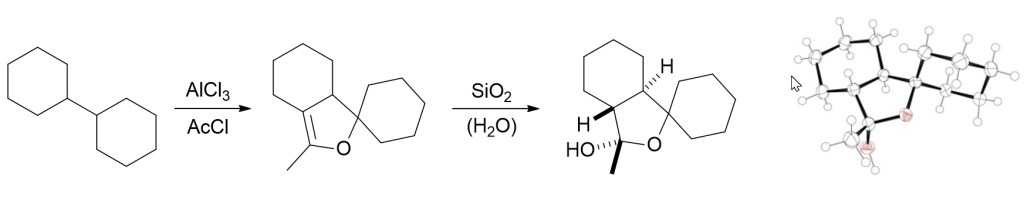
19.) “Aliphatic C–H Activation with Aluminium Trichloride–Acetyl Chloride: Expanding the Scope of the Baddeley Reaction for the Functionalisation of Saturated Hydrocarbons”, Lyall, C. L.; Uosis-Martin, M.; Lowe, J. P.; Mahon, M. F.; Pantoş, G. D.; Lewis, S. E.* Org. Biomol. Chem., 2013, 11, 1468, doi:10.1039/c2ob26765a
2012
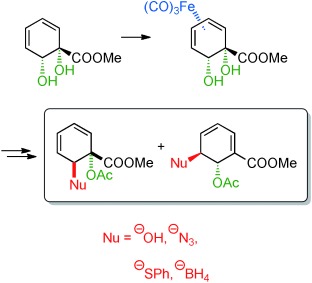
18.) “Valuable New Cyclohexadiene Building Blocks via Cationic η5 Iron Carbonyl Complexes Derived from a Microbial Arene Oxidation Product”, Ali Khan, M.; Mahon, M. F.; Lowe, J. P.; Stewart, A. J. W.; Lewis, S. E.* Chem. Eur. J., 2012, 18, 13480, doi:10.1002/chem.201202411
17.) “Photooxygenation of a Microbial Arene Oxidation Product and Regioselective Kornblum–DeLaMare Rearrangement. Total Synthesis of Zeylenols and Zeylenones”, Palframan, M. J.; Kociok-Köhn, G.; Lewis, S. E.* Chem. Eur. J., 2012, 18, 4766, doi:10.1002/chem.201104035
2011
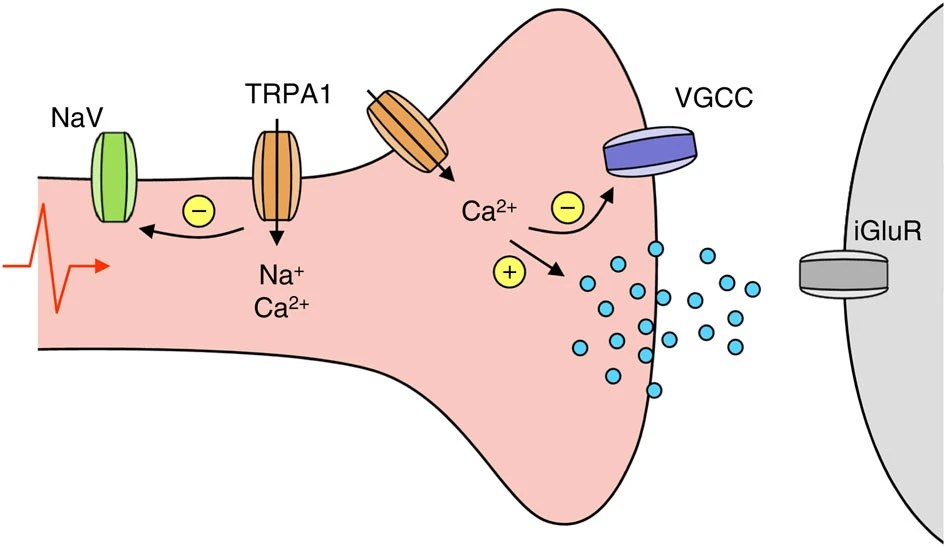
16.) “TRPA1 Mediates Spinal Antinociception Induced by Acetaminophen and the Cannabinoid Δ9-Tetrahydrocannabiorcol”, Andersson, D. A.; Gentry, C.; Alenmyr, L.; Killander, D.; Lewis, S. E.; Andersson, A.; Bucher, B.; Galzi, J.-L.; Sterner, O.; Bevan, S.; Högestätt, E. D.; Zygmunt, P. M. Nature Commun., 2011, 2, 551, doi:10.1038/ncomms1559
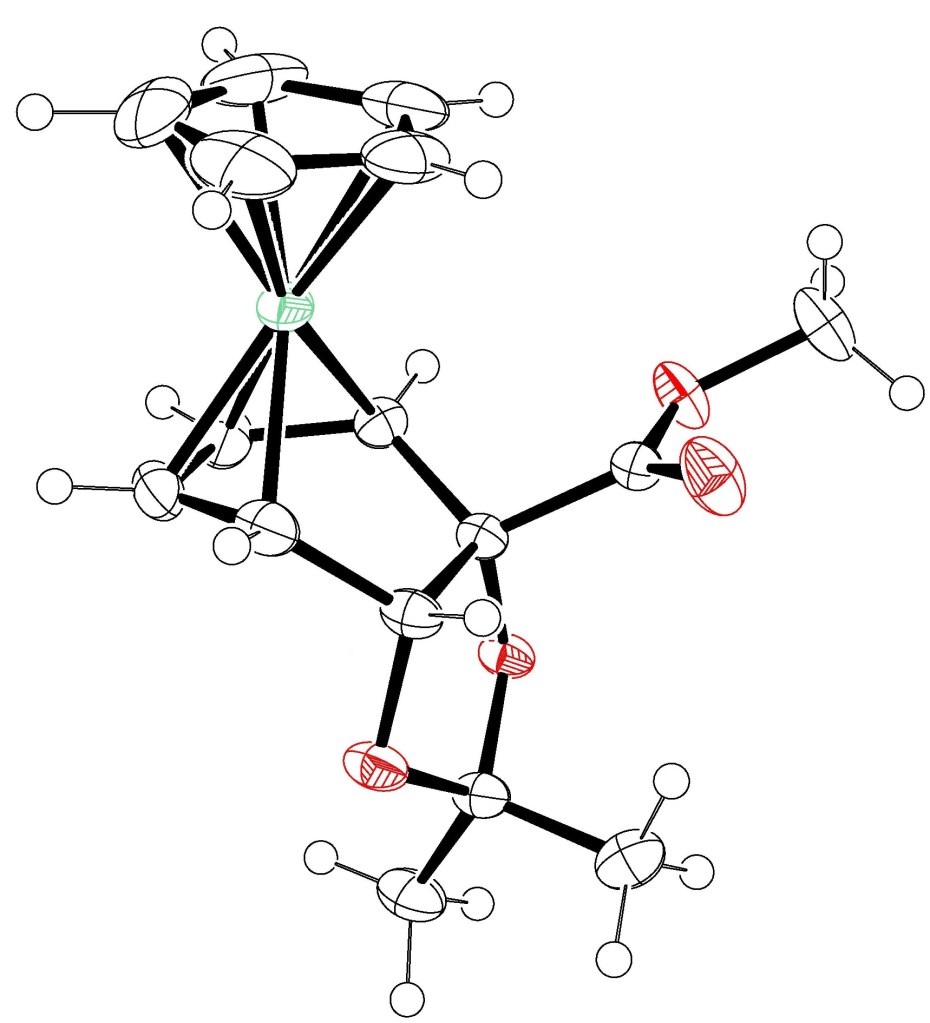
15.) “A Cobalt Complex of a Microbial Arene Oxidation Product”, van der Waals, D.; Pugh, T.; Ali Khan, M.; Stewart, A. J. W.; Johnson, A. L.; Lewis, S. E.* Chem. Central J., 2011, 5, 80, doi:10.1186/1752-153X-5-80
14.) “Concise Synthesis of 1,4a-Bifunctionalised Decalin Building Blocks by C–H Activation of Decalin”, Uosis-Martin, M.; Mahon, M. F.; Yevglevskis, M.; Lewis, S. E.* Synlett, 2011, 2211, doi: 10.1055/s-0030-1261184
13.) “Synthetic methods Part (II): oxidation and reduction methods”, Asghar, S. F.; Lewis, S. E.* Annu. Rep. Prog. Chem., Sect. B: Org. Chem., 2011, 107, 34–67, doi:10.1039/c1oc90012a [Not open access]
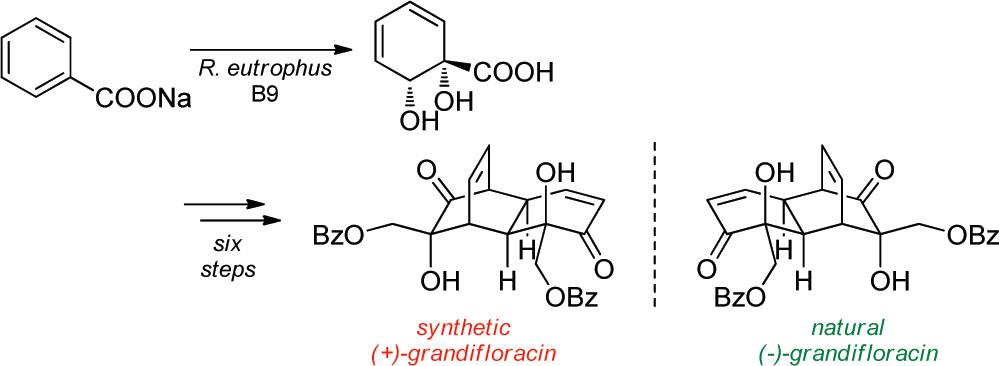
12.) “Total Synthesis of (+)-Grandifloracin by Iron Complexation of a Microbial Arene Oxidation Product”, Palframan, M. J.; Kociok-Köhn, G.; Lewis, S. E.* Org. Lett., 2011, 13, 3150, doi:10.1021/ol201057r
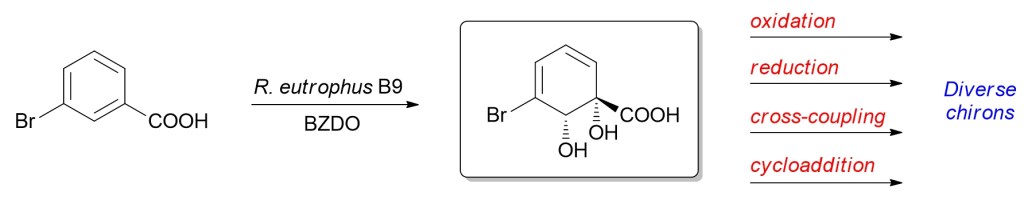
11.) “Expanding the chiral pool: oxidation of meta-bromobenzoic acid by R. eutrophusB9 allows access to new reaction manifolds”, Griffen, J. A.; Le Coz, A. M.; Kociok-Köhn, G.; Ali Khan, M.; Stewart, A. J. W; Lewis, S. E.* Org. Biomol. Chem., 2011, 9, 3920, doi:10.1039/c1ob05131h
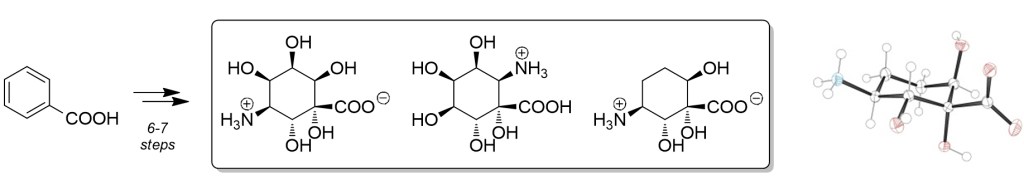
10.) “Inosaminoacids: novel inositol–amino acid hybrid structures accessed by microbial arene oxidation”, Pilgrim, S.; Kociok-Köhn, G.; Lloyd, M. D.; Lewis, S. E.* Chem. Commun., 2011, 47, 4799, doi:10/1039/c1cc10643k
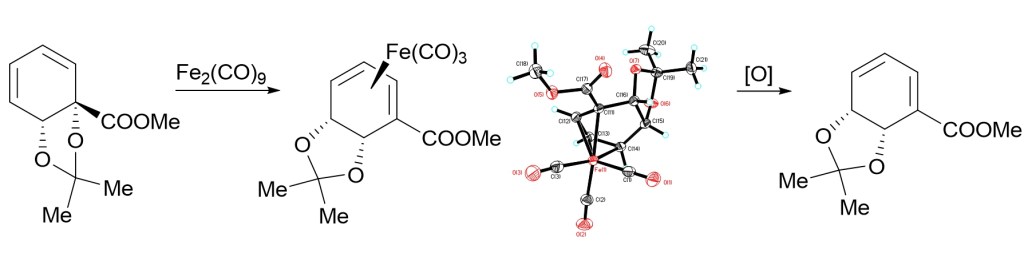
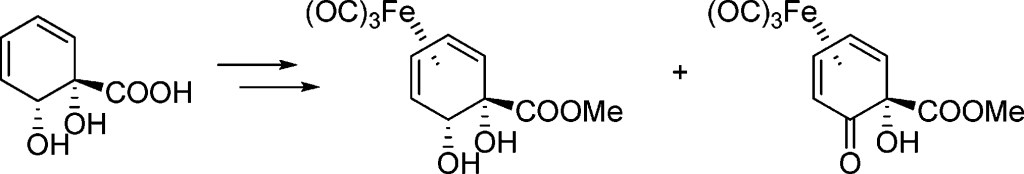
9.) “Accessing the antipodal series in microbial arene oxidation: a novel diene rearrangement induced by tricarbonyliron(0) complexation“, Ali Khan, M.; Lowe, J. P.; Johnson, A. L.; Stewart, A. J. W.; Lewis, S. E.* Chem. Commun., 2011, 47, 215, doi:10.1039/c0cc01169j
2010
8.) “Iron(0)tricarbonyl Complexes of Microbially-Derived Cyclohexadiene Ligands Containing Quaternary Stereocenters”, Ali Khan, M.; Mahon, M. F.; Stewart, A. J. W.; Lewis, S. E.* Organometallics, 2010, 29, 199, doi:10.1021/om9009069 [Not open access]
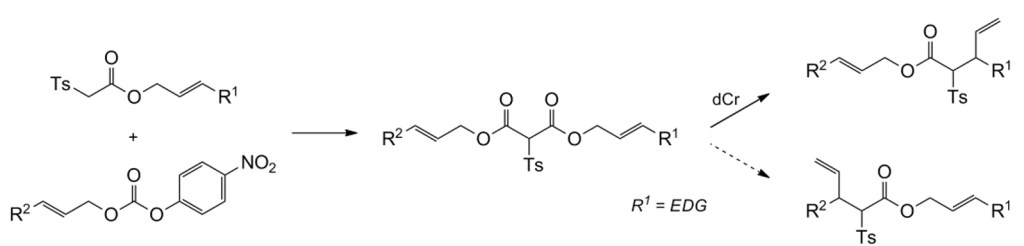
7.) “Transannular decarboxylative Claisen rearrangement reactions for the synthesis of sulfur-substituted vinylcyclopropanes”, Craig, D.*; Gore, S. J.; Lansdell, M. I.; Lewis, S. E.; Mayweg, A. V. M.; White, A. J. P. Chem. Commun., 2010, 46, 4991, doi:10.1039/c0cc00976h [Not open access]
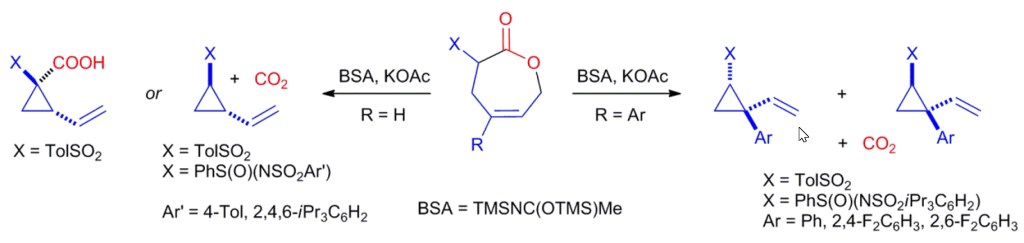
6.) “Synthesis and Characterisation of Novel Alkane-α,ω-diyl bis(silyl triflates)”, Baker, T.; Lewis, S. E.* Synth. Commun., 2010, 40, 2747, doi:10.1080/00397910903318724 [Not open access]
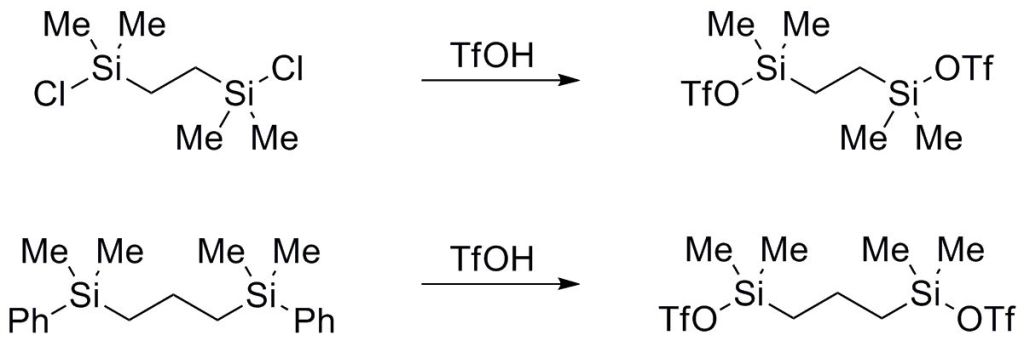
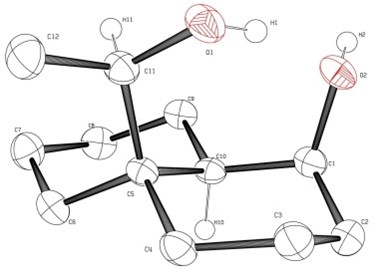
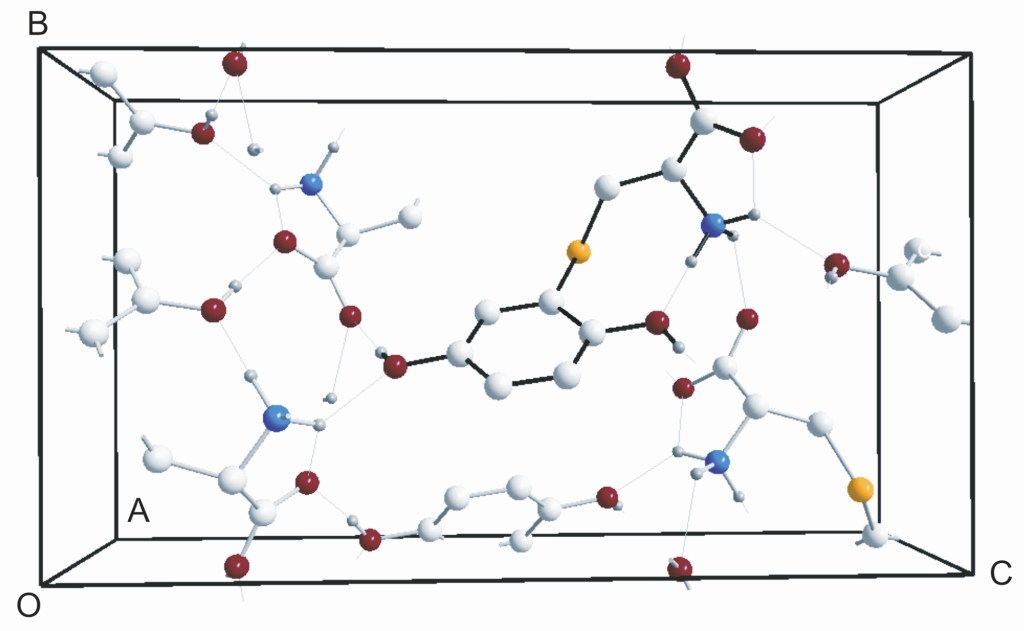
5.) “Crystallographic rationalization of the reactivity and spectroscopic properties of (2R)-S-(2,5-dihydroxyphenyl)cysteine”, Kociok-Köhn, G.; Lewis, S. E.* Acta Cryst., 2010, C66, o187, doi:10.1107/S0108270110005780
4.) “Synthetic methods: Part (ii) Oxidation and reduction methods”, Lewis, S. E.* Annu. Rep. Prog. Chem., Sect. B: Org. Chem., 2010, 106, 34–75. doi:10.1039/b927087f [Not open access]
2009
3.) “Synthetic methods: Part (ii) Oxidation and reduction methods”, Lewis, S. E.* Annu. Rep. Prog. Chem., Sect. B: Org. Chem., 2009, 105, 35–74. doi:10.1039/b822050f [Not open access]
2007
2.) “Decarboxylative Claisen rearrangements of diallyl 2-sulfonylmalonates: remarkable regioselectivity in the reaction of bifunctional substrates”, Craig, D.*; Lansdell, M.; Lewis, S. E. Tetrahedron Lett.,2007, 48, 7861–7864. doi:10.1016/j.tetlet.2007.08.130 [Not open access]
2006
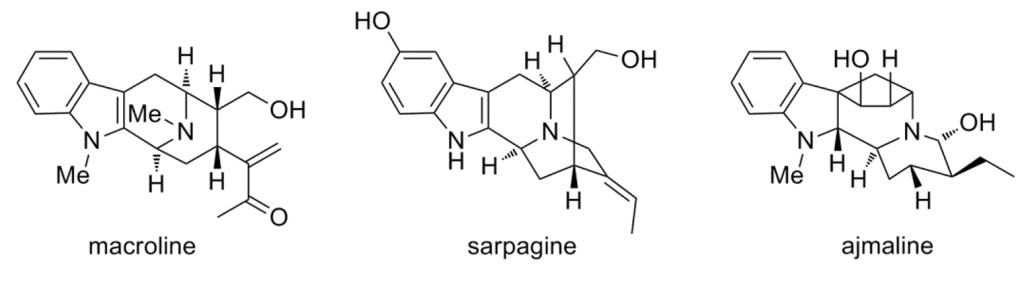
1.)“Recent advances in the chemistry of macroline, sarpagine and ajmaline-related indole alkaloids”, Lewis, S. E.* Tetrahedron, 2006, 62, 8655–8681. doi:10.1016/j.tet.2006.06.017 [Not open access]